
Home
Studies
& Data Analysis
Methods
Microscope studies
Flagella experiment
Laboratory math
Blood fractionation
Gel electrophoresis
Protein gel analysis
Mitochondria
Concepts/ theory
Keeping a lab notebook
Writing research papers
Dimensions & units
Using figures (graphs)
Examples of graphs
Experimental error
Representing error
Applying statistics
Principles of microscopy
Solutions & dilutions
Protein assays
Spectrophotometry
Fractionation & centrifugation
Radioisotopes and detection
Guide to the study
Lab part 1
- tutorial/specimens
- Paramecium
- Chlamydomonas
- fixing/observingflagella
- Chaos (Pelomyxa) carolinensis
- Naegleria gruberi
- the five kingdoms
Lab part 2
- experiment introduction
- microtubules
- amputating flagella
- experimental design
- data collection
- class data
Chaos (Pelomyxa) carolinensis
Chaos is a member of the phylum Sarcodina, consisting of ameoba-like organisms. The Sarcodina are heterotrophs, that is, for energy they rely on photosynthetic organisms either directly or indirectly. Their principal means of obtaining nutrients is by ingestion. Chaos are primarily scavengers, found on the bottom of freshwater habitats. However, they do ingest other living organisms, such as Paramecium.
You may picture protozoa as too small to be seen without a microscope. True, many protozoa are only a few micrometers in diameter, but some species such as Chaos carolinensis are macroscopic, that is, visible to the unaided eye. To observe living Chaos you will need to prepare a vaseline mount. If concentrated Paramecium are available, you might add a drop to the preparation so that you can see them captured in the food vacuoles. Careful! Chaos are big enough to crush with a coverslip.
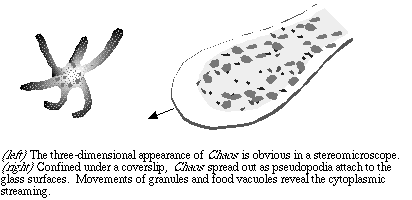
Chaos are too big to be examined at high power, unless you can concentrate on a very thin pseudopodium. To start, center an amoeba so that you know it is in the field of view. In bright field mode use the lowest power available on your microscope, and focus. Although the ameoba is unstained, it is thick enough to be obvious in bright field as long as your provide reasonable contrast with the aperture diaphragm. Swing in the 10x objective lens (100x total magnification) to see more detail.
Examine the specimen in dark field, which may better reveal the granules. As the specimen attaches to the glass and begins to move you may find rapidly moving pseudopodia. Switch to the high dry (400 or 430x magnification) and center the end of a growing pseudopod in the field of view. Try using dark field and phase contrast to see the details.
Now that you've had a look at Chaos you might wish to make some organized observations. The protist may appear to be completely disorganized at first, but close examination of the pseudopodia reveals that the cytoplasm is subdivided into two compartments. The endoplasm is quite fluid and contains the multiple nuclei, granules, and food vacuoles. The peripheral part of the cytoplasm, the ectoplasm, is much more viscous and has no granules. As pseudopodia grow the distinction is very apparent. Part of the currently accepted model for amoeboid motion is the transformation of the gel-like state of the ectoplasm to the more fluid state of the endoplasm during cytoplasmic movements. That is, the cytoplasm can transform between a viscous plasmagel and a fluid plasmasol.
To describe how amoeboid motion takes place, two models have been proposed, one of which is relatively recent. The tail contraction model was proposed by S. O. Mast in 1926. Mast's model suggested that force is generated in the uroid (tail) region of the amoeba, so that the gel-like ectoplasm pushes the fluid endoplasm toward the tip of the pseudopod. The endoplasm is advanced passively, and the membrane is forced to expand.
Consider the Mast model for a moment. Can you identify a uroid region in Chaos? By examining the cytoplasmic movements, can you tell if the endoplasm advances passively or is simply pushed?
Another model was proposed by Allen and Taylor in 1975. In the frontal contraction model, a contractile force is generated at the site of the hyaline cap (the ectoplasm-containing tip of the growing pseudopodium). The endoplasm is actually pulled forward rather than being pushed. Can you support or reject this model, based on your visual observations?
Use your microscopic skills to study the patterns of advancing and retreating pseudopodia. Observe the initiation of movement in pseudopodia by watching the movement of granules. Do you think it is possible to confirm one or the other model using microscopic observation alone? Let's put it this way... would you bet your career on one model or the other without further evidence?
While relatively few organisms use amoeboid motion for locomotion, many organisms rely on the process for specific tissue functions or at specific stages. For example, amoeboid motion is a part of some developmental processes. Human white blood cells, specifically monocytes and granulocytes, can respond to signals by carrying out diapedesis, that is, using amoeboid motion to leave the blood and squeeze into tissues between the cells of capillaries. Of far more importance, however, is the similarity between amoeboid motion and cytoplasmic streaming in general. Cytoplasmic movements play an essential role in the function of most cell types, including cells of sessile organisms (most plants). The same molecular mechanisms that are responsible for pseudopod development in Chaos may be involved in the organization of most of our own cells into functional tissues.
Chaos and other amoebae can be useful models for study of the mechanisms behind cytoplasmic streaming. We believe that actin microfilaments are involved in generating the required force, perhaps by treadmilling (lengthening at one end while shortening at the other), or by interaction with myosin in a manner similar to muscle contraction. Understanding the mechanisms requires identification of the different types of actin, myosin, and other microfilaments as well as associated proteins. It involves studying how they can interact and how their interactions can be regulated. Questions such as this are complex and are beyond the ability of any single scientist to answer. Yet knowledge of how such movements occur is critical to our development of a full understanding of cellular processes. Clearly, investigations into mechanisms such as this require far more information than can be provided by simple microscopic observation.
Experiment: ingestion of Paramecium by Chaos
Cultures of Chaos can be maintained for a long time in the laboratory as long as the water is replaced regularly and the cultures are fed. We feed them Paramecium, either by adding concentrated Paramecium in order to produce large numbers of amoebae in a short time, or to maintaining cultures by creating a food chain with Chaos at the top (we just "seed" a Paramecium culture with a few Chaos, and the critters take care of the rest). Considering the speed of amoeboid versus cililary motion, one might wonder how the slow-moving amoebae capture the Paramecium in the first place. Here is how we set up a wet mount to observe the process.
One or two Chaos amoebae were removed from culture with a 9 in. pasteur pipet, with the aid of a dissecting microscope to locate individual cells. The amoebae were placed in the center of a clean glass microscope slide, with a small volume of medium. A small drop of concentrated Paramecium was added, and a vaseline mount prepared. A thick ledge of vaseline was used and the coverslip pressed down just sufficiently to contact the medium and spread the drop. Ameobae were then examined at low magnifications (40x, 100x) and the process of ingestion of Paramecium was observed.
Excessive pressure was found to crush the amoebae, and predator became prey. If the pressure of the coverslip was sufficient to cause the amoeba to begin to spread, the damage was done.
If you try this experiment, try to come up with a possible explanation for the ease with which the amoebae capture the faster-moving Paramecium.
Visitors: to ensure that your message is not mistaken for SPAM, please include the acronym "Bios211" in the subject line of e-mail communications
Created by David R. Caprette (caprette@rice.edu), Rice University 7 Sept 95
Updated 13 Aug 07
