
Home
Studies
& Data Analysis
Methods
Microscope studies
Flagella experiment
Laboratory math
Blood fractionation
Gel electrophoresis
Protein gel analysis
Mitochondria
Concepts/ theory
Keeping a lab notebook
Writing research papers
Dimensions & units
Using figures (graphs)
Examples of graphs
Experimental error
Representing error
Applying statistics
Principles of microscopy
Solutions & dilutions
Protein assays
Spectrophotometry
Fractionation & centrifugation
Radioisotopes and detection
Introduction/
training
Mitochondria theory
Mitochondria in vitro
- preparation
- fate of substrates
- state IV
- state III
- metabolic poisons
- mitotraces
- rationale
- experiments
Additional topics
Overview of Mitochondria Structure and Function
The organelles we call mitochondria are found in the cytoplasm of nearly all eukaryotic cells. Their most immediate function is to produce adenosine triphosphate (ATP) by systematically extracting energy from nutrient molecules (substrates). ATP is the universal energy-yielding commodity in cells, used by enzymes to perform a wide range of cellular functions. We cannot survive, even for a moment, without a sufficient supply of ATP.
We refer to the process as aerobic metabolism because it requires constant removal of excess electrons through the reduction of oxygen. The need for oxygen as an electron acceptor is the sole reason that we breathe air. In fact, the most immediate purpose of our respiratory and circulatory systems is to deliver oxygen to the tissues for use by mitochondri (and to eliminate carbon dioxide). By the way, the term respiration is synonymous with the phrase "cellular respiration." It refers to the reduction of oxygen to water. Simply inhaling and exhaling is simply breathing, not respiration.Enzymes and energetics
In order to understand the role of mitochondria in cells, you will need some very basic concepts concerning how cells store and transfer energy. All molecules contain energy, stored in the molecular structure itself. A portion of that energy, called free energy, can be used to do work. A chemical reaction that adds free energy to a molecule is said to reduce the molecule. Removing free energy from a molecule is called oxidation. When a reaction results in transfer of free energy from one molecule to another we call it an oxidation/reduction, or redox reaction. In a redox reaction one or more molecules is reduced (gains energy) while one or more molecules is oxidized (loses energy).
In cells, enzymes are responsible for reactions that transfer free energy from one molecule to another. A typical enzyme is extremely selective for its substrates (the reactants). Enzymes bind substrates in such a way that they are brought in to positions favoring a particular reaction, greatly lowering the activation energy for the reaction. By controlling enzyme activity a cell can control what reactions take place, because the types of reactions catalyzed by enzymes are extremely unlikely to occur spontaneously.
The second law of thermodynamics states that spontaneous reactions occur in directions that increase the overall disorder of the universe. A consequence is that with each energy transfer some energy is lost to the chaotic motion of molecules that we measure as temperature. While enzymes are designed to conserve free energy, some energy is always 'wasted' with each process (although endotherms use the 'wasted' energy to maintain body temperature). The stepwise oxidation of substrates by enzymes can be thought of as a 'controlled burn,' in which much of the available free energy is retained in a useful form.
Sources of nutrients
When we discuss metabolism, we refer to reactants as substrates. Enzymes can act upon many kinds of fatty acids, sugars, and amino acids. Various metabolic pathways 'channel' the molecules into a limited number of forms, which we call Krebs substrates (after Hans Krebs, who is credited with the discovery of the basic metabolic cycle within mitochondria). Conversion to Krebs substrates is accomplished in the cytoplasm, in the mitochondria matrix, or in both places, depending on the reaction. A limited number of enzymes need be maintained in the mitochondria, and energy from a variety of sources is funneled into what ultimately becomes a single pathway.
Structure
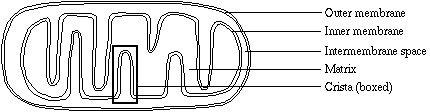
Krebs' cycle

At certain points in the cycle, carbon dioxide is lopped off, so the number of carbon atoms from citrate to oxaloacetate is reduced from 6 to 4. Hydrogens are removed when water molecules are taken off of the substrates. So the system is in balance. But what happens to the free energy that is being released by progressive oxidation of substrates?
Energy carriers
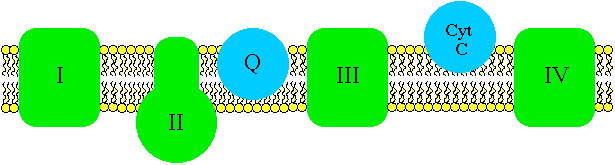
Energy carriers in turn lose energy to molecules that are embedded in the inner membrane. They are then reoxidized and ready to be used again (note that FAD is part of complex II in the inner membrane). The purpose of the carriers is to shuttle energy from the Krebs reactions to specific structures in the inner membrane. The molecules embedded in the inner membrane are energy carriers of a different type. They transfer energy by passing along a pair of electrons. Taken together, the electron-transporting molecules in the inner membrane are called the electron transport system.
Electron transport
Components of the electron transport system include complexes I, II, III, and IV, plus two individual molecules, coenzyme Q and cytochrome c.
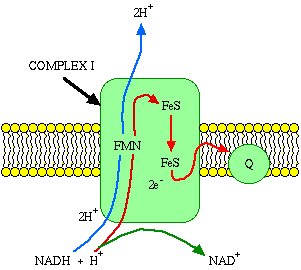
NADH is reoxidized at complex I. A pair of electrons from NADH is then passed through a series of electron transport carriers within complex I to coenzyme Q (NOT to complex II). The electrons flow just like water rolling downhill, with energy released upon each electron exchange. Within complex I, as each electron pair passes through a pair of hydrogen ions (protons) is forced from the matrix to the intermembrane space.
Electrons cannot be passed through unless the protons are translocated at the same time. The energy used to force the protons across the inner membrane is released by the passage of the electron pair from NADH to coenzyme Q. The electrons are then passed through complex III, where more protons are forced across, and complex IV, where still more protons are forced across. Complex IV, called cytochrome oxidase, uses the energy it receives along with an electron pair to reduce oxygen to water. Note that since molecular oxygen is diatomic it takes two cytochrome oxidase complexes, two electron pairs, and four hydrogen ions to complete the reaction.
Oxygen must be replenished, otherwise electron transport cannot proceed. Each carrier, once reduced, would have to stay that way because there would be no place for the electrons to go. The need to deliver oxygen to the electron transport system is why we have respiratory and circulatory systems. Oxygen is necessary to "drain" electrons from the system, otherwise all of the carriers would remain reduced and electron transport would have to stop.
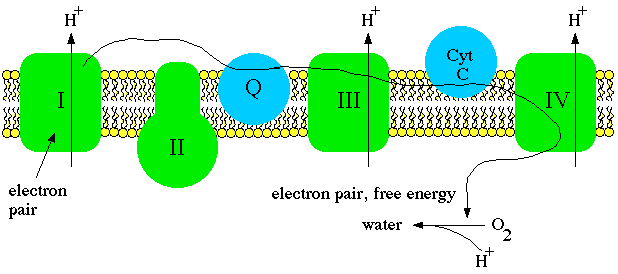
As electrons are tranferred from carrier to carrier, the total amount of free energy drops. Each carrier is more electronegative than the carrier "upstream." Much of the energy is lost as heat, but a significant amount is stored in the form of a proton gradient.
Chemiosmotic gradient and respiratory control
The electron transport system cannot force an infinite number of protons into the intermembrane space. The inner membrane is impermeable to protons, and they accumulate in the intermembrane space, creating what is called a chemiosmotic gradient. A mitochondrion in vivo maintains its energy gradient at a constant level. That situation does not change because of a mechanism that we call respiratory control.
Electron transport cannot proceed if protons cannot be pumped across the inner membrane. Protons cannot be pumped unless the available energy to move them out of the matrix exceeds the required amount plus what energy is lost to heat. If the inner and outer membranes were sealed and there was no way of altering the gradient, electron transport would have to stop. In reality, a number of processes act constantly to dissipate the gradient, so electron transport never completely stops. Instead, the rate of electron transport is regulated by the rate at which the energy is removed from the gradient. The ability of the chemiosmotic gradient to limit electron transport is called respiratory control.
Many students have trouble understanding the cause and effect relationship between chemiosmotic gradient and electron transport. Nevertheless it is the most critical concept toward a complete understanding of how mitochondria function.
Oxidative phosphorylation
Embedded in the inner membrane among the structures of the electron transport system are structures called the ATP synthase complex. The complex consists of a proton channel and catalytic sites for the synthesis of ATP from ADP and phosphate. When ADP and phosphate are available, they bind the catalytic sites on the ATP synthase. When this happens, the channel opens, and protons can come whooshing back in. The energy released is used to couple the phosphate to ADP, to make ATP.
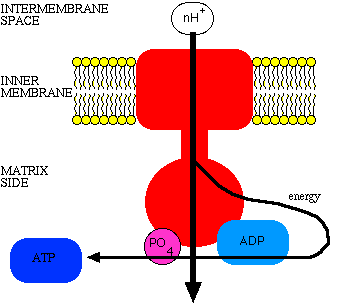
The mechanism can be likened to a water wheel, where the flow of protons resembles a flow of water downhill, and the turning of the wheel is the turning of ADP toward phosphate to cause the bond to form.
Stimulation of electron transport by ADP
When ADP binds to ATP synthase, protons are driven into the matrix and as they do so energy is drained from the gradient. As fast as energy is removed, the electron transport system can force protons into the intermembrane space. The faster or slower energy is removed from the gradient, the faster or slower electron transport can proceed. Again, the rate of electron transport is subject to respiratory control.
Summary of key concepts
- Reactions that remove free energy are oxidation reactions; reactions that add free energy are reduction reactions
- Enzymes facilitate oxidation and reduction reactions at the same time, by transferring free energy from one molecule to another; some energy is always lost in such transfers
- Locations: Krebs reactions take place in the matrix; electron transport takes place on the inner membrane; a gradient is maintained across the inner membrane; ATP synthesis takes place in the matrix as protons cross the inner membrane from the intermembrane space
- Nutrients from a variety of sources are modified so that mitochondrial metabolism involves a limited number of Krebs substrates
- Metabolic products can enter Krebs cycle in a number of places
- Krebs reactions transfer energy from the progressive oxidation of substrates to two types of energy carriers, NAD and FAD
- Energy carriers donate electrons and free energy to the electron transport system (ETS)
- Each successive carrier in the ETS, when reduced, carries less available free energy than the one upstream
- Electron transport cannot take place without pumping protons out of the matrix
- Proton pumping produces a chemiosmotic gradient that in turn restricts the rate of electron transport
- Various processes drain energy from the gradient, allowing electron transport to proceed
- The most important process that exploits the gradient is ATP synthesis
- Mitochondria in vivo constantly exercise respiratory control
- Electron transport always proceeds as fast as energy is removed from the gradient
Visitors: to ensure that your message is not mistaken for SPAM, please include the acronym "Bios211" in the subject line of e-mail communications
Created by David R. Caprette (caprette@rice.edu), Rice University 12 Dec 96
Updated 23 Aug 07
