
Home
Studies
& Data Analysis
Methods
Microscope studies
Flagella experiment
Laboratory math
Blood fractionation
Gel electrophoresis
Protein gel analysis
Mitochondria
Concepts/ theory
Keeping a lab notebook
Writing research papers
Dimensions & units
Using figures (graphs)
Examples of graphs
Experimental error
Representing error
Applying statistics
Principles of microscopy
Solutions & dilutions
Protein assays
Spectrophotometry
Fractionation & centrifugation
Radioisotopes and detection
- overview; types; bright field microscopy
- dark field optics
- phase contrast
- oil immersion
- differential interference contrast
- measuring
- using a counting chamber
- wet (Vaseline) mount
Dark Field Viewing
Dark field optics are a low cost alternative to phase contrast optics. The contrast and resolution obtained with inexpensive dark field equipment may be superior to what you have with student grade phase contrast equipment. It is surprising that few manufacturers and vendors promote the use of dark field optics.
A dilute suspension of yeast cells makes a good practice specimen for dark field optics, particularly when cultured with living Paramecium.
Principle
To view a specimen in dark field, an opaque disc is placed underneath the condenser lens, so that only light that is scattered by objects on the slide can reach the eye (figure 2). Instead of coming up through the specimen, the light is reflected by particles on the slide. Everything is visible regardless of color, usually bright white against a dark background. Pigmented objects are often seen in "false colors," that is, the reflected light is of a color different than the color of the object. Better resolution can be obtained using dark field as opposed to bright field viewing.
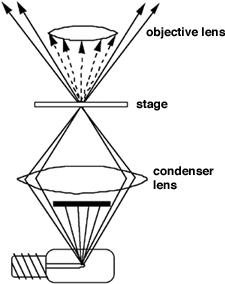
You don't need sophisticated equipment to get a dark field effect, although the effect is most dramatic when the occulting disk is built into the condenser itself. You do need a higher intensity light, since you are seeing only reflected light. At low magnification (up to 100x) any decent optical instrument can be set up so that light is reflected toward the viewer rather than passing through the object directly toward the viewer.
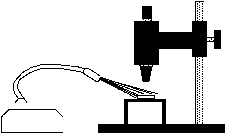
To set up a dissecting microscope for "dark field" viewing, the specimen should be placed over an opening so that light reflects only from surfaces between cover slip and slide, not from a surface beneath the slide. You may need to make a stand to hold the slide. The surface beneath the opening should be a flat black. Turn off any built-in illuminator. Aim a high-intensity light source toward the specimen at an angle, from the top or side through a glass dish or jar.
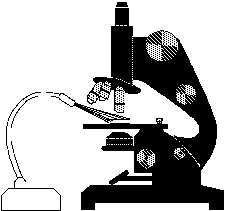
With a compound microscope, dark field is obtained by placing an occulting disk in the light path between source and condenser. A cheap set of occulting disks can be prepared by cutting circular pieces of black electrical tape ranging from dime-size up to a diameter that equals the width of the slide, and sticking them to the slide in a row. The circles should be spaced well apart. A specimen is placed on the microscope stage as usual, and the illumination should be made as uniform as possible. If there is an aperture diaphragm in the condenser (contrast lever), it should be opened up wide. After focusing at low power, the slide with occulting disks is placed in the light path between source and condenser, bringing it as close to the bottom of the condenser as it will go.
I would start with the largest disk, sliding it around until it is directly in the center of the light path. Increasing the illumination should then produce a good dark field effect. To optimize, first try stopping down the field diaphragm to get the best contrast between background and specimen. Try to match the size of the occulting disk to the field diameter, so that the edge of the disk is just outside the field of veiw - smaller disks are appropriate for higher power objectives. Vertically, the disk should be a close to the condenser as possible, to make the contrast the greatest. On microscopes with built-in dark field equipment, the view is so impressive because the occulting disk is built into the condenser - very close and focused. After testing the set-up this way, a stand might be rigged to fit under the microscope, so the slide can be placed in position without holding it. Something that 'grabs' the condenser and supports the occulting disks would be ideal. The less the students have to mess with, the better.
I set this up on the crummiest little piece of garbage microscope I could find, and it looked very good. A relatively new student-model microscope should give a much better effect.
Suspensions of cells and samples of pond water look spectacular in dark field. While specimens may look washed out and lack detail in bright field, protists, metazoans, cell suspensions, algae, and other microscopic organisms are clearly distinguished and their details show up well. At 100x you can readily see bacteria, even distinguish some structure (rods, curved rods, spirals, or cocci) and movement. Non-motile bacteria look like vibrating bright dots against a dark background. Motile bacteria can be seen moving in a definite direction, sometimes remarkably fast. In pond water samples you may find Spirillum volutans, a very large (up to 0.5 mm) motile spiral bacterium.
When to use dark field illumination
Dark field illumination is most readily set up at low magnifications (up to 100x), although it can be used with any dry objective lens. Any time you wish to view everything in a liquid sample, debris and all, dark field is best. Even tiny dust particles are obvious. Dark field is especially useful for finding cells in suspension. Dark field makes it easy to obtain the correct focal plane at low magnification for small, low contrast specimens. Use dark field for
-
Initial examination of suspensions of cells such as yeast, bacteria, small protists, or cell and tissue fractions including cheek epithelial cells, chloroplasts, mitochondria, even blood cells (small diameter of pigmented cells makes it tricky to find them sometimes despite the color).
-
Initial survey and observation at low powers of pond water samples, hay or soil infusions, purchased protist or metazoan cultures.
-
Examination of lightly stained prepared slides. ? Initial location of any specimen of very small size for later viewing at higher power.
-
Determination of motility in cultures
Visitors: to ensure that your message is not mistaken for SPAM, please include the acronym "Bios211" in the subject line of e-mail communications
Created by David R. Caprette (caprette@rice.edu), Rice University 11 MAY 00
Updated 10 Aug 12
