Bizarre
Results Explained
Bizarre – example 1
The large container of glycine looked the same
as the large container of choline chloride. Choline
is a lot more hygroscopic than glycine, which should
have been a clue that the wrong component was used
in the electrode buffer. Choline is a basic compound
and certainly does not substitute for glycine.
We caught the error and replaced the buffer after
electrophoresis had proceeded for some time, but
it was too late for this one.
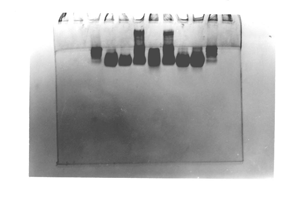
Bizarre – example 2
This one was kindly contributed by
T. Sedlacek, Czech Republic. It was run with high
current and overheated.

Bizarre – example 3
Here are two gels that were contributed
by E. Morales Rayas. A likely explanation is that
each time there was a delay between loading the
samples and actually running the gel. The middle
lanes of the first gel show an alternating pattern
in which every other lane is either wide or compressed.
Bands on the second gel spread out toward the edges.
When samples sit in a well the proteins
begin to diffuse into the stacking gel, both vertically
and laterally. Smaller proteins diffuse more rapidly
than do larger ones. If proteins in a sample diffuse
laterally they may alter the electric field affecting
adjacent lanes, especially if the samples in adjacent
wells contain predominantly higher mass polypeptides.
The problem isn't so bad when all of the samples
are of similar composition (second gel) and the
lanes are loaded in sequence. However distortion
still occurs.
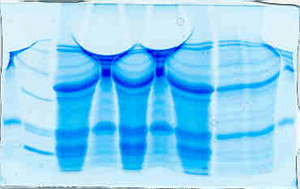  
Bizarre – example 4
Evidently it is not a good idea to
store tris-glycine electrode buffer in a gallon
plastic bottle on a shelf in the lab. We didn't
see any mold in the buffer, but by golly it was
there, all right. Notice that in each example the
gel below the dye front was cleared following destaining.
The persistent blue background is from mold proteins
that continued to penetrate the gel throughout
the procedure.
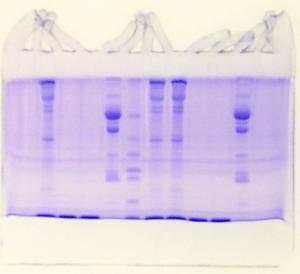  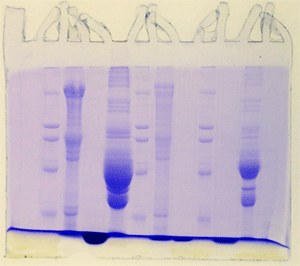
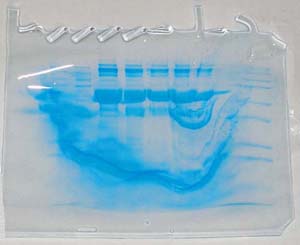
Bizarre – example 5
Here's one thing that can happen
if someone opens a gel box before the run is completed,
WITH THE POWER ON! Another thing that can happen
is that the bozo can get killed! This one was contributed
by Margaret A. Buttery, Notre Dame De Namur University
in Belmont, California.
|


![]()













