Smeared Protein Gels
Smeared gels – example 1
Smearing can have a variety of causes, but most commonly
it is due to an unevenly poured acrylamide mixture
or due to gross overloading of protein.
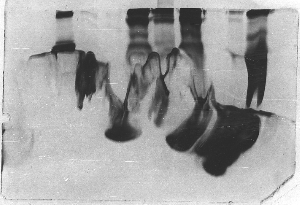
In this example the gel was not properly poured,
so that the lower half had begun to polymerize
before the upper part was poured. The first gel
mix began to polymerize too quickly. Rather than
prepare a new gel cassette, the students simply
stopped pouring, prepared a new mix, and poured
it on top of the old. Obviously, the bond wasn't
particularly good.
Smeared gels – example 2
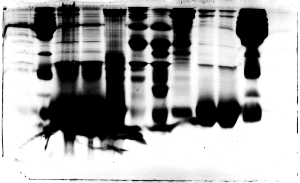
In this example a lot of protein was loaded in
each of the wells, all the way across. Most of
the lanes can still be interpreted, but smearing
is particularly evident in lanes 3 and 4. Those
lanes contained primarily one protein (hemoglobin
subunit), as did lanes 9 and 10. However, the group
that loaded 3 and 4 underestimated the protein
concentrations in their red cell lysate and red
cell cytoplasm fractions. Those fractions contained
so much protein that samples had to be diluted
prior to preparing a protein assay tube. The students
forgot to take the dilution factor into consideration
when determining protein concentration.
The capacity of a mini-gel for a mixed protein
sample is 20 to 40 micrograms/well, depending on
the resolution needed and number individual polypeptides
in the mix. However, if a pure protein is loaded,
one single band will contain all 20-40 micrograms,
and the result is a mess.
Smeared gels – example 3
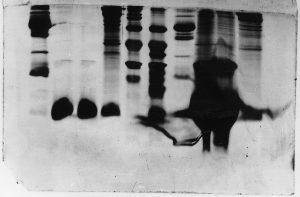
Here is another example of gross overloading, and
it appears these students made the same mistake as
in example 2. It looks as though there was some inconsistency
in the gel also.
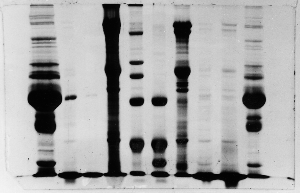
Smeared gels – example 4
Smearing can be a normal consequence of running
membrane-associated proteins with a high lipid
content in a discontinuous gel. In discontinuous
PAGE, sample proteins are super-concentrated by
a combination of two factors. First, the rate of
migration is suddenly slowed as the sample moves
from a non-restrictive stacking gel to a separating
gel that restricts movement. Second (and more important),
a pH change affects the electrophoretic mobility
of proteins in the sample, causing the sample to
check up abruptly. A sample of a half cm or so
in height becomes compressed into a layer of bands
a few micrometers thick, and the local concentration
of protein goes up considerably.
Membrane-associated proteins tend to precipate
at lower concentrations, thus lanes containing
such proteins often have a dark band of precipitated
proteins at the very top. As electrophoresis proceeds
the precipates re-dissolve and enter the gel continuously,
thus forming a continuous dark background of unresolved
polypeptides. Many of the membrane samples shown
on these gels will show that characteristic.
The resolved bands in the membrane protein lanes
(4 and 7) appear to consist of similar amounts
of protein, however in lane 4 the capacity of the
gel for at least some of the membrane proteins
was exceeded, resulting in a dark smear.
|


![]()










