Nanoparticle-Assembled Capsules as Advanced Encapsulation/Delivery Agents.

SEM image of tin oxide NACs (average diameter = 620 nm) prepared from SnO2 NPs, poly(allylamine), and phosphate anions.
There are several methods to prepare microcapsule structures: surfactant self-assembly, sacrificial templating, and physical extrusion. Vesicles or liposomes are hollow sphere structures composed of a bilayer of surfactant molecules. They can form very easily and can readily encapsulate water-soluble compounds; however, the structure is very sensitive to fluid conditions and is difficult manipulate physically. In sacrificial templating, solid or liquid particles are enveloped by a solid shell, and the inner particles are removed through solvent extraction, chemical dissolution, or high temperatures. Uniformly sized shells result from this multi-step process, but non-destructive encapsulation is difficult to perform. Physical extrusion encompasses many methods for the scalable generation of coated liquid droplets, in which the coating contains a crosslinkable precursor that reacts or hardens around the droplet (that contains the target encapsulate). Shell sizes smaller than 1 micron are difficult to achieve, though.
Uniquely different from these methods is polymer aggregate templating, which is the simplest method yet to generate stable microcapsule structures for encapsulation purposes. This materials synthesis route is quite general, as a variety of NP compositions and polymers can be used. The nanoscale properties of the constituent NPs can be incorporated into the NACs, and the polymer molecular structure can be modified to a great extent (to increase functionality, for example) as long as the charge-driven NP assembly process is not disrupted.
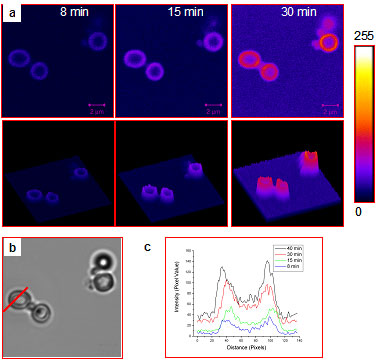
Representative microcapsules prepared from citrate-bridged PLL aggregates and SiO2 NPs that contain acid phosphatase enzyme,
in an aqueous solution containing fluorescein diphosphate: (a) a series of time-lapse confocal microscopy images collected over the course of 30 min, (b) brightfield
image, and (c) line intensity profiles along the red line shown in (b). The reactant diffuses inside the microcapsules and interacts with the encapsulated enzyme to
form the fluorescent fluorescein product. Scale bars: 2 micrometers.
The mild conditions (room temperature, atmospheric pressure, near-neutral pH, water solvent) and simplicity that allow microcapsule synthesis to be a highly scalable process, also allow compounds to be encapsulated easily and without damage. The encapsulation of dye molecules, drug compounds, and enzymes, for example, can be carried out by simply contacting the target compounds with the polymer aggregates prior to shell formation (i.e., prior to addition of the shell-forming NPs).
Enzyme-containing NACs can be considered individual micro-bioreactors. The enzymes are protected by the NAC shell wall from direct contact with the external aqueous environment, but can interact with reactant molecules that penetrate the semipermeable shell wall via concentration- gradient-driven diffusion. There is no well-defined pore size or shape in the shell walls, but it is presumed that the transport pathway is defined by the interstitial spaces of the NP/polymer composite.
Selected Publications
N.X. Zhao, H.G. Bagaria, M.S. Wong, and Y.L. Zu, "A nanocomplex that is both tumor cell-selective and cancer gene-specific for anaplastic large cell lymphoma," J Nanobiotechnol, 9, 2 (2011). DOI:10.1186/1477-3155-9-2 (Abstract)
G.C. Kini, S.L. Biswal, and M.S. Wong, "Non-LBL Assembly and Encapsulation Uses of Nanoparticle-Shelled Hollow Spheres," Adv Polym Sci, 229, 175-200 (2010). DOI:10.1007/12_2010_53 (Abstract) (Link)
J. Yu, D. Javier, M.A. Yaseen, N. Nitin, R. Richards-Kortum, B. Anvari and M.S. Wong, "Self-assembly Synthesis, Tumor Cell Targeting, and Photothermal Capabilities of Antibody-coated Indocyanine Green Nanocapsules," J. Am. Chem. Soc., 132 (6), 1929-1938 (2010). DOI:10.1021/ja908139y (Abstract)
S.E. Plush, M. Woods, Y. Zhou, S.B. Kadali, M.S. Wong and A.D. Sherry, "Nanoassembled Capsules as Delivery Vehicles for Large Payloads of High Relaxivity Gd3+ Agents",J. Am. Chem. Soc., 131 (43), 15918–15923 (2009). DOI:10.1021/ja906981w (Abstract)
P.R. LeDuc, M.S. Wong, et al., "Towards an in vivo Biologically Inspired Nanofactory",Nature Nanotech., 2, 3-7 (2007). DOI:10.1038/nnano.2006.180 (Abstract)