2009 Abstracts.
57. W. Y. L. Ko, H. G. Bagaria, S. Asokan, K.-J. Lin and M. S. Wong, "CdSe Tetrapod Synthesis Using Cetyltrimethylammonium Bromide and Heat Transfer Fluids," J. Mater. Chem. 20(12), 2474-2478 (2010) DOI:10.1039/b922145j

The synthesis of CdSe tetrapod-shaped quantum dots using phenyl-based heat transfer fluids as inexpensive alternatives to octadecene solvent was studied. The CdSe tetrapods were synthesized using the hot-injection method, in which the trioctylphosphine selenide precursor and the shape-inducing cetyltrimethylammonium bromide surfactant were injected into a cadmium oleate-containing solvent at 190 °C. At a synthesis temperature of 160 °C, the resulting quantum dot particles were found to grow more slowly in heat transfer fluids and pure phenyl-type solvents than in octadecene. With synthesis time, the selectivity to tetrapods increased, and the arms grew proportionally in width and length. The use of heat transfer fluids provides a convenient means to control growth of shaped nanoparticles.
56. W. Zhou, E. I. Ross-Medgaarden, W. V. Knowles, M. S. Wong, I. E. Wachs and C. J. Kiely, "Identification of Active Zr–WOx Clusters on a ZrO2 Support for Solid Acid Catalysts" Nature. Chem 1, 722-728 (2009). DOI:10.1038/NCHEM.433
Tungstated zirconia is a robust solid acid catalyst for light alkane (C4–C8) isomerization. Several structural models for catalytically active sites have been proposed, but the topic remains controversial, partly because of the absence of direct structural imaging information on the various supported WOx species. High-angle annular dark-field imaging of WO3/ZrO2 catalysts in an aberration-corrected analytical electron microscope allows, for the first time, direct imaging of the various species present. Comparison of the relative distribution of these WOx species in materials showing low and high catalytic activities has allowed the deduction of the likely identity of the catalytic active site—namely, subnanometre Zr–WOx clusters. This information has subsequently been used in the design of new catalysts, in which the activity of a poor catalyst has been increased by two orders of magnitude using a synthesis procedure that deliberately increases the number density of catalytically relevant active species.
55. S.E. Plush, M. Woods, Y. Zhou, S.B. Kadali, M.S. Wong and A.D. Sherry, "Nanoassembled Capsules as Delivery Vehicles for Large Payloads of High Relaxivity Gd3+ Agents" J. Am. Chem. Soc., 131(43), 15918–15923 (2009). DOI:10.1021/ja906981w
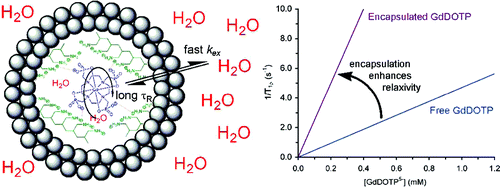
Nanoassembled capsules (NACs) that incorporate a polymer aggregate inside a silica shell may be loaded with agents that are of particular interest for therapeutic or diagnostic applications. NACs formed using the MRI contrast agent GdDOTP in the internal polymer aggregate are reported herein, the smaller of which show promise as potential MRI contrast agents. Unlike many other nanoencapsulated systems, water access to the inner core of these NACs does not appear to be limited and consequently the water relaxivity per Gd3+ agent can reach as high as 24 mM s-1. Robust, spherical capsules were formed using polyallylamine or poly-l-lysine ranging from 0.2 to 5 mm in diameter. The greatest gains in relaxivity were observed for smaller NACs, for which water accessibility remained high but molecular rotation of the Gd3+ chelate was effectively restricted. Larger NACs did not afford such large gains in relaxivity, the result of poorer water accessibility combined with less-effective rotational restriction.
54. M. A. Yaseen, J. Yu, B. Jung, M. S. Wong, and B. Anvari, "Biodistribution of Encapsulated Indocyanine Green in Healthy Mice" Mol. Pharmaceutics, online (2009). DOI: 10.1021/mp800270t
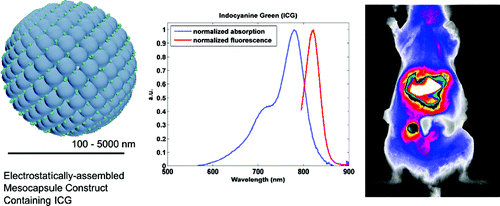
Indocyanine green (ICG) is a fluorescent probe used in various optically mediated diagnostic and therapeutic applications. However, utility of ICG remains limited by its unstable optical properties and nonspecific localization. We have encapsulated ICG within electrostatically assembled mesocapsules (MCs) to explore its potential for targeted optical imaging and therapy. In this study, we investigate how the surface coating and size of the MCs influences ICG’s biodistribution in vivo. ICG was administered intravenously to Swiss Webster mice as a free solution or encapsulated within either 100 nm diameter MCs coated with dextran; 500 nm diameter MCs coated with dextran; or 100 nm diameter MCs coated with 10 nm ferromagnetic iron oxide nanoparticles, themselves coated with polyethylene glycol. ICG was extracted from harvested blood and organs at various times and its amount quantified with fluorescence measurements. MCs containing ICG accumulated in organs of the reticuloendothelial system, namely, the liver and spleen, as well as the lungs. The circulation kinetics of ICG appeared unaffected by encapsulation; however, the deposition within organs other than the liver suggests a different biodistribution mechanism. Results suggest that the capsules’ coating influences their biodistribution to a greater extent than their size. The MC encapsulation system allows for delivery of ICG to organs other than the liver, enabling the potential development of new optical imaging and therapeutic strategies.
53. K. N. Heck, M. O. Nutt, P. Alvarez, and M. S. Wong, "Deactivation Resistance of Pd/Au Nanoparticle Catalysts for Water-phase Hydrodechlorination" J. Catal. 267, 97-104 (2009) DOI:10.1016/j.jcat.2009.07.015
Palladium-decorated gold nanoparticles (Pd/Au NPs) have recently been shown to be highly efficient for trichloroethene hydrodechlorination, as a new approach in the treatment of groundwater contaminated with chlorinated solvents. Problematically, natural groundwater can contain chloride and sulfide ions, which are known poisons in Pd-based catalysis. In this study, the effects of chloride and sulfide on the trichloroethene hydrodechlorination catalytic activity were examined for non-supported Pd/Au NPs and Pd NPs, and alumina-supported Pd (Pd/Al2O3). Over the concentration range of 0-0.02 M NaCl, the catalytic activity of Pd/Au NPs was unaffected, while the activities of both the Pd NPs and Pd/Al2O3 catalyst dropped by ~70%. Pd/Au NPs were found to be highly resistant to sulfide poisoning, deactivating completely at a ratio of sulfide to surface Pd atom (S:Pdsurf) of at least 1, compared to Pd NPs deactivating completely at a ratio of 0.5. Pd/Al2O3 retained activity at a ratio of 0.5, pointing to a beneficial role of the Al2O3 support. Sulfide poisoning of Pd/Au NPs with different Pd surface coverages provided a way to assess the nature of active sites. The gold component was found to enhance both Pd catalytic activity and poisoning resistance for room-temperature, water-phase trichloroethene hydrodechlorination.
The effects of chloride and sulfide on water-phase trichloroethene hydrodechlorination using Pd-on-Au nanoparticles (Pd/Au NPs), Pd NPs, and alumina-supported Pd were studied. Pd/Au NPs were resistant to chloride poisoning unlike monometallic Pd, and they showed greater resistance to sulfide poisoning than monometallic Pd. Lower Pd content surface coverages led to less activity but resistance to sulfide poisoning.
52. E. Muñoz Tavera, S. B. Kadali, H. G. Bagaria, A. W. Liu,and M. S. Wong ,"Experimental and Modeling Analysis of Diffusive Release from Single-shell Microcapsules" AIChE J. DOI:10.1002/aic.11914
There is much experimental and mathematical work that describes chemical transport from multilayered films of planar geometries. There is less so, however, for chemical transport from multilayered spheres, a common structure for controlled-release materials. Based on the Sturm-Liouville approach of Ramkrishna and Amundson (1974), explicit analytical solutions for the concentration profiles and release kinetics from spherical capsules are presented. Fluorescent dye-release studies using single-shelled microspheres called nanoparticle-assembled capsules were performed to validate the model for uniformly and nonuniformly sized capsules. The combined experiment-modeling approach allows optical microscopy images and release measurements to be readily analyzed for estimating diffusion coefficients in capsule core and shell walls.
51. V.S. Murthy, S.B. Kadali, and M.S. Wong, "Polyamine-Guided Synthesis of Anisotropic, Multicompartment Microparticles" Appl. Mater. Interfac., 1, 590-596 (2009). DOI:10.1021/am8001499
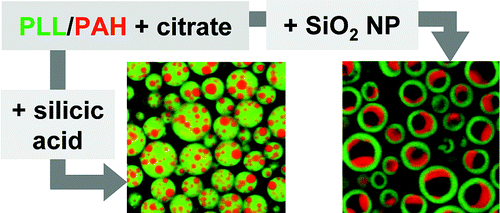
Colloidal particles that have nonuniform bulk or surface compositions are of emerging interest because of their potential applications involving advanced chemical storage and delivery and the self-assembly of novel functional materials. Experimental realization of anisotropic particles is much more difficult than that for particles with uniform bulk and surface composition, however. A new wet-chemical synthesis method to anisotropic microparticles is presented. This approach makes convenient use of the unusual observation of a salt-triggered separation of two water-solubilized polyamines into colloidal aggregates with nonuniform polymer composition. The anisotropic structure of these ionically cross-linked aggregates is explained by the difference in surface tensions of the contained single-polymer domains. Contacting the polymer aggregates with silicic acid or 13-nm silica nanoparticles leads to the charge-driven formation of solid or hollow microspheres, respectively. Depending on the poly(lysine)/poly(allylamine) ratio, the nonuniformity of the polymer aggregates translates to surface patches or internal compartments found in the resultant silica/polymer microparticles. Such hybrid materials with their unique structure could serve as a new basis for targeted chemical delivery and controlled release for potential applications in medicine, food, and cosmetics.
50. E. I. Ross-Medgaarden, I. E. Wachs, W. V. Knowles, A. Burrows, C. J. Kiely, and M. S. Wong, "Tuning the Electronic and Molecular Structure of Catalytic Active Sites with Titania Nanoligands" J. Am. Chem. Soc., 131, 680-687 (2009). DOI:10.1021/ja711456c

A series of supported 60% TiO2/SiO2 catalysts were synthesized and subsequently used to anchor surface VOx redox and surface WOx acid sites. The supported TiOx, VOx, and WOx phases were physically characterized with TEM, in situ Raman and UV-vis spectroscopy, and chemically probed with in situ CH3OH-IR, CH3OH-TPSR and steady-state CH3OH dehydration. The CH3OH chemical probe studies revealed that the surface VOx sites are redox in nature and the surface WOx sites contain acidic character. The specific catalytic activity of surface redox (VO4) and acidic (WO5) sites coordinated to the titania nanoligands are extremely sensitive to the degree of electron delocalization of the titania nanoligands. With decreasing titania domain size, 10 nm, acidic activity increases and redox activity decreases due to their inverse electronic requirements. This is the first systematic study to demonstrate the ability of oxide nanoligands to tune the electronic structure and reactivity of surface metal oxide catalytic active sites.
49. M.S. Wong, P. J.J. Alvarez, Y.L. Fang, N. Akcin, M. O. Nutt, J. T. Miller, and K. N. Heck, "Cleaner Water using Bimetallic Nanoparticle Catalysts" J. Chem. Tech. & Biotech, 84, 158-166 (2009). DOI:10.1002/jctb.2002
Groundwater contaminated by hazardous chlorinated compounds, especially chlorinated ethenes, continues to be a significant environmental problem in industrialized nations. The conventional treatment methods of activated carbon adsorption and air-stripping successfully remove these compounds by way of transferring them from the water phase into the solid or gas phase. Catalysis is a promising approach to remove chlorinated compounds completely from the environment, by converting them into safer, non-chlorinated compounds. Palladium-based materials have been shown to be very effective as hydrodechlorination catalysts for the removal of chlorinated ethenes and other related compounds. However, relatively low catalytic activity and a propensity for deactivation are significant issues that prevent their widespread use in groundwater remediation. Palladium-on-gold bimetallic nanoparticles, in contrast, were recently discovered to exhibit superior catalyst activity and improved deactivation resistance. This new type of material is a significant next-step in the development of a viable hydrodechlorination catalysis technology.