2010 Abstracts.
68. L. A. Pretzer, Q. X. Nguyen, and M. S. Wong, "Controlled Growth of Sub-10 nm Gold Nanoparticles Using Carbon Monoxide Reductant," J. Phys. Chem. C, 114 (49), 21226-21233 (2010). DOI: 10.1021/jp107945d
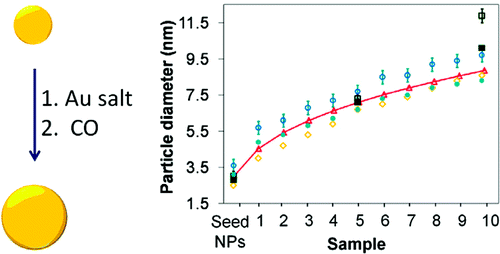
There is a need to develop aqueous-phase synthesis methods of sub-10-nm Au nanoparticles (NPs), given their many exciting possibilities in catalysis, sensing, biomedical, and other water-based applications. Synthesizing Au NPs by reducing Au salt onto preformed NPs as seeds is a useful approach because final particle size can be finely predicted and controlled, though few studies have reported successful synthesis of Au NPs in the 1 and 10 nm size range. Here we report that water-suspended Au particles with a diameter ~2.8 nm can be grown as large as ~12 nm with sub-nanometer control, as verified through detailed ultraviolet?visible spectroscopy, small-angle X-ray scattering, and transmission electron microscopy measurements. With carbon monoxide as the reducing agent, this seeded-growth method results in colloidally stable Au sols. The reaction mechanism most likely involves the catalyzed oxidation of CO into CO2 accompanied by electron transfer to the gold hydroxide-chloride ionic species through the growing particle.
67. H. G. Bagaria, G. C. Kini, and M. S. Wong, "Electrolyte Solutions Improve Nanoparticle Transfer from Oil to Water," J. Phys. Chem. C, 114 (47), 19901-19907 (2010). DOI: 10.1021/jp106140j
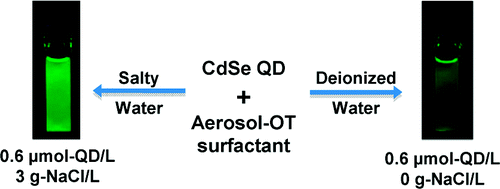
Many techniques to transfer NPs from the oil phase into the water phase have been developed, but all have limitations that remain to be addressed, specifically low transfer yields and partial NP aggregation. One of the transfer processes involves emulsifying an oil suspension of NPs in water with surfactants, followed by evaporation of the oil to produce water suspensions of surfactant-encapsulated NPs. We show here that using salt solutions instead of deionized water significantly improves this emulsion-based transfer process. With oleate-coated CdSe quantum dots as a model NP system, hexane as the oil phase, sodium bis(2-ethylhexyl) sulfosuccinate (AOT) as the emulsifying surfactant, and NaCl solution as the water phase, the resultant aqueous suspensions exhibit months-long photoluminescence stability, near 100% phase-transfer yield, and nonaggregation. These benefits are attributed to a more laterally packed AOT layer surrounding the NP, as supported by zeta-potential measurements, surface-charge calculations, thermogravimetry, and Nile Red fluorescence analysis. The new phase-transfer method is general for a variety of NP, surfactant, and salt types.
66. J. Yu, J. M. Berlin, W. Lu, L. Zhang, A. T. Kan, P. Zhang, E. E. Walsh, S. N. Work, W. Chen, J. M. Tour, M. S. Wong, and M. B. Tomson, "Transport study of nanoparticles for oilfield application," SPE 131158, 2010 SPE International Conference on Oilfield Scale, Aberdeen, UK, May 26-27., (online) DOI: 10.2118/131158-MS
Recently, the revolution of nanotechnology has been noticed for its many potential applications in the oil & gas industry such as enhanced oil recovery process and subsurface mapping. Understanding the transport and retention of nanoparticles (NPs) in an oilfield environment, such as high salinity, high temperature, high pressure, and heterogeneous pore distribution is critical to their application. The objective of this study is to investigate the fundamental transport and retention properties of NPs in the challenging oilfield conditions. In this work, dolomite core material from an oil well in Kuwait and Berea sandstone were used. Carbon NPs were used as model nanomaterials. The core sample was washed with toluene to remove residual oil, ground to 106 – 250 ?m grains, and packed into columns. Carbon NP breakthrough curves were collected in synthetic seawater at room temperature. The results showed that the existence of salt ions dramatically delayed NP breakthrough time and increased NP retention. Effects of different types of salts and salt concentrations are discussed. NP transport appears to be highly dependent on the degree of interaction between the NPs, salt ions in solution, and porous medium surface, which affects retardation and retention. NP breakthrough can be significantly improved by avoiding this interaction via NP surface modification. Our preliminary results provided helpful guidelines for NP transport in oil & gas applications. The results are discussed in detail. Furthermore, the experimental results were fitted with a 1-D advective and dispersion equation combined with a first-order removal term to obtain important transport parameters for NPs that did not aggregate. More complicated models are being developed to capture the unusual breakthrough curves associated with NP aggregation.
64. N. Soultanidis, W. Zhou, A. C. Psarras, A. J. Gonzalez, E. F. Iliopoulou, C. J. Kiely, I. E. Wachs, and M. S. Wong, "Relating n-Pentane Isomerization Activity to the Tungsten Surface Density of WOx/ZrO2," J. Am. Chem. Soc., 132 (38), 13462-13471 (2010). DOI: 10.1021/ja105519y
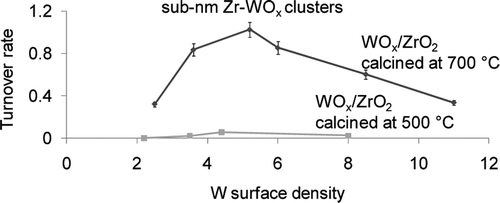
Zirconia-supported tungsten oxide (WOx/ZrO2) is considered an important supported metal oxide model acid catalyst, for which structure-property relationships have been studied for numerous acid-catalyzed reactions. The catalytic activity for xylene isomerization, alcohol dehydration, and aromatic acylation follows a volcano-shape dependence on tungsten surface density. However, WOx/ZrO2 has not been studied for more acid-demanding reactions, like n-pentane isomerization, with regard to surface density dependence. In this work, WOx/ZrO2 was synthesized using commercially available amorphous ZrOx(OH)4-2x and model crystalline ZrO2 as support precursors. They were analyzed for n-pentane isomerization activity and selectivity as a function of tungsten surface density, catalyst support type, and calcination temperature. Amorphous ZrOx(OH)4-2x led to WOx/ZrO2 (WZrOH) that exhibited maximum isomerization activity at 5.2 W·nm-2, and the crystalline ZrO2 led to a material (WZrO2) nearly inactive at all surface densities. Increasing the calcination temperature from 773 to 973 K increased the formation of 0.8-1 nm Zr-WOx clusters detected through direct imaging on an aberration-corrected high-resolution scanning transmission electron microscope (STEM). Calcination temperature further increased catalytic activity by at least two times. Brønsted acidity was not affected but Lewis acidity decreased in number, as quantified via pyridine adsorption infrared spectroscopy. WOx/ZrO2 exhibited isomerization activity that peaked within the first 2 h time-on-stream, which may be due to Zr-WOx clusters undergoing an activation process.
63. J. A. Jamison, E. L. Bryant, S. B. Kadali, M. S. Wong, V. L. Colvin, K. Matthews, and M. K. Calabretta, "Altering Protein Surface Charge with Chemical Modification Modulates Protein-Gold Nanoparticle Aggregation," J. Nanopart. Res., 13 (2), 625-636 (2010). DOI:10.1007/s11051-010-0057-5
Gold nanoparticles (AuNP) can interact with a wide range of molecules including proteins. Whereas significant attention has focused on modifying the nanoparticle surface to regulate protein–AuNP assembly or influence the formation of the protein “corona,” modification of the protein surface as a mechanism to modulate protein–AuNP interaction has been less explored. Here, we examine this possibility utilizing three small globular proteins—lysozyme with high isoelectric point (pI) and established interactions with AuNP-lactalbumin with similar tertiary fold to lysozyme but low pI; and myoglobin with a different globular fold and an intermediate pI. We first chemically modified these proteins to alter their charged surface functionalities, and thereby shift protein pI, and then applied multiple methods to assess protein–AuNP assembly. At pH values lower than the anticipated pI of the modified protein, AuNP exposure elicits changes in the optical absorbance of the protein–NP solutions and other properties due to aggregate formation. Above the expected pI, however, protein–AuNP interaction is minimal, and both components remain isolated, presumably because both species are negatively charged. These data demonstrate that protein modification provides a powerful tool for modulating whether nanoparticle–protein interactions result in material aggregation. The results also underscore that naturally occurring protein modifications found in vivo may be critical in defining nanoparticle–protein corona compositions.
62. J. Yu, D. Javier, M. A. Yaseen, N. Nitin, R. Richards-Kortum, B. Anvari and M.S. Wong, "Self-assembly Synthesis, Tumor Cell Targeting, and Photothermal Capabilities of Antibody-coated Indocyanine Green Nanocapsules," J. Am. Chem. Soc., 132(6), 1929-1938 (2010). DOI:10.1021/ja908139y
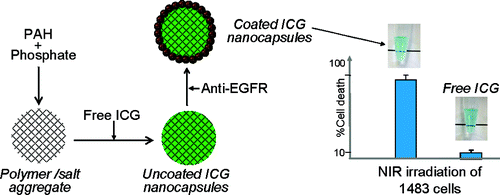
New colloidal materials that can generate heat upon irradiation are being explored for photothermal therapy as a minimally invasive approach to cancer treatment. The near-infrared dye indocyanine green (ICG) could serve as a basis for such a material, but its encapsulation and subsequent use are difficult to carry out. We report the three-step room-temperature synthesis of 120-nm capsules loaded with ICG within salt-cross-linked polyallylamine aggregates, and coated with antiepidermal growth factor receptor (anti-EGFR) antibodies for tumor cell targeting capability. We studied the synthesis conditions such as temperature and water dilution to control the capsule size and characterized the size distribution via dynamic light scattering and scanning electron microscopy. We further studied the specificity of tumor cell targeting using three carcinoma cell lines with different levels of EGFR expression and investigated the photothermal effects of ICG containing nanocapsules on EGFR-rich tumor cells. Significant thermal toxicity was observed for encapsulated ICG as compared to free ICG at 808 nm laser irradiation with radiant exposure of 6 W/cm2. These results illustrate the ability to design a colloidal material with cell targeting and heat generating capabilities using noncovalent chemistry.
61. G. C. Kini, S. L. Biswal, and M. S. Wong, "Non-LBL Assembly and Encapsulation Uses of Nanoparticle-Shelled Hollow Spheres," Adv Polym Sci, 229, 175-200 (2010). DOI:10.1007/12_2010_53
Nanoparticles (NPs, diameter range of 1–100nm) can have size-dependent physical and electronic properties that are useful in a variety of applications. Arranging them into hollow shells introduces the additional functionalities of encapsulation, storage, and controlled release that the constituent NPs do not have. This chapter examines recent developments in the synthesis routes and properties of hollow spheres formed out of NPs. Synthesis approaches reviewed here are recent developments in the electrostatics-based tandem assembly and interfacial stabilization routes to the formation of NP-shelled structures. Distinct from the well-established layer-by-layer (LBL) synthesis approach, the former route leads to NP/polymer composite hollow spheres that are potentially useful in medical therapy, catalysis, and encapsulation applications. The latter route is based on interfacial activity and stabilization by NPs with amphiphilic properties, to generate materials like colloidosomes, Pickering emulsions, and foams. The varied types of NP shells can have unique materials properties that are not found in the NP building blocks, or in polymer-based, surfactant-based, or LBL-assembled capsules.
60. T.-C. Tseng, E. S. McGarrity, J. W. Kiel, P. M. Duxbury, M. E. Mackay, A. L. Frischknecht, S. Asokan, and M. S. Wong, "Three-dimensional Liquid Surfaces through Nanoparticle Selfassembly," Soft Matter, 6, 1533-1538 (2010).DOI:10.1039/b918429e

Nanoparticles blended into thin polymer films are driven to assemble at interfaces by both entropic and enthalpic forces. When nanoparticles assemble at a substrate–polymer film interface, these forces are so large that they enable the film to follow surface protrusions and form a three-dimensional surface, instead of dewetting from the substrate. In other words, disassembling the nanoparticle layer requires more energy than that gained by the film dewetting from the rough surface. Here we studied blends of linear polystyrene and CdSe nanoparticles spin coated onto silicon substrates containing sparsely distributed SiO2 particles (ca. 120 nm diameter). The films were then thermally annealed for periods of up to 24 h, well above the glass transition temperature of the polymer. The profiles of different film thicknesses (40–180 nm) were characterized using atomic force microscopy (AFM) before and after being annealed and were found to become much smoother, yet remain three-dimensional after annealing with a profile that gradually decayed away from the SiO2 particles. Calculations based on a continuum theory using a balance of assembly, dispersion and surface tension forces were performed and found to be in agreement with the AFM profile data demonstrating the strength of the assembly forces.
59. W. Y. L. Ko, H. G. Bagaria, S. Asokan, K.-J. Lin and M. S. Wong, "CdSe Tetrapod Synthesis Using Cetyltrimethylammonium Bromide and Heat Transfer Fluids," J. Mater. Chem. 20(12), 2474-2478 (2010) DOI:10.1039/b922145j

The synthesis of CdSe tetrapod-shaped quantum dots using phenyl-based heat transfer fluids as inexpensive alternatives to octadecene solvent was studied. The CdSe tetrapods were synthesized using the hot-injection method, in which the trioctylphosphine selenide precursor and the shape-inducing cetyltrimethylammonium bromide surfactant were injected into a cadmium oleate-containing solvent at 190 °C. At a synthesis temperature of 160 °C, the resulting quantum dot particles were found to grow more slowly in heat transfer fluids and pure phenyl-type solvents than in octadecene. With synthesis time, the selectivity to tetrapods increased, and the arms grew proportionally in width and length. The use of heat transfer fluids provides a convenient means to control growth of shaped nanoparticles.
58. G. C. Kini, J. Lai, M. S. Wong, and S. L. Biswal, "Microfluidic Formation of Ionically Cross-Linked Polyamine Gels," Langmuir (online) DOI:10.1021/la903983y
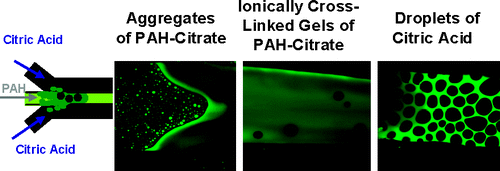
In this article, we discuss in situ polymer gelation in microfluidic channels from electrostatically mediated interactions when reactant streams of a linear cationic polymer (poly(allylamine hydrochloride, PAH) and a multivalent anion (sodium citrate) are subjected to shear flow. We find that the polyamine exhibits shear-thickening behavior as it is ionically cross-linked by citrate ions to form viscoelastic gel phases. These gels form at room temperature and remain stable and intact after the cessation of flow. Gelation is found to occur in the polymer stream and not the citrate stream because of an appreciably higher diffusivity of citrate ions when compared to the gel and PAH and because of laminar flow conditions in the microfluidic environment. Gel formation occurred when the pH of the PAH stream was below the PAH pKa value of 8.38 and when citrate was either in a disodium or trisodium state. The formation of aggregates, gels, and droplets was found to depend strongly on the charge ratio and flow conditions. The gelation of PAH begins with the formation of colloidal aggregates of PAH and citrate, which then combine under shear flow to form noncontinuous or continuous gels. Droplets of citrate can form within regions of continuous gels as excess citrate anions diffuse into the gel stream.