Nanoparticle Assembly.
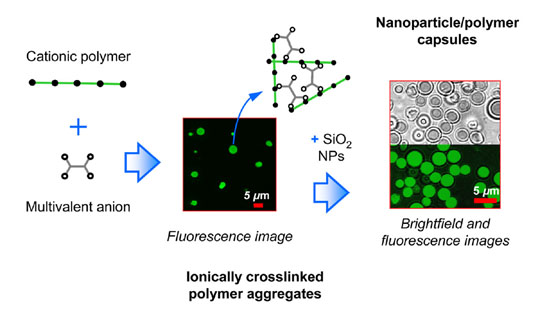
Schematic of NP assembly: polymer aggregate templating

Optical (differential interference contrast) image of a microcapsule suspension.
With the advances made in solution-phase materials chemistry in the last 20 years, nanoparticles (NPs) can now be prepared out of a wide spectrum of compositions with a high degree of particle size and shape control. NPs are an intriguing class of materials because their reduced physical dimensionality leads to the appearance of catalytic, chemical, optoelectronic, and magnetic properties not found in bulk materials. They remain rather difficult to handle for applications, though, due to their colloidal nature and their susceptibility to uncontrolled aggregation. Thus, there is a need for synthetic methodologies for creating functional materials out of NPs.
We recently discovered that, under specific solution conditions, cationic polyelectrolytes can induce negatively-charged silica NPs to form micron-sized hollow spheres rather than the randomly structured precipitate that would ordinarily result from flocculation. The polyelectrolyte (e.g., polyallylamine) forms aggregates under the crosslinking action of a multivalent salt (e.g., EDTA). These aggregates act as templates around which the NPs deposit to form a multilayer-thick NP/polymer shell. We term this form of NP assembly as "polymer aggregate templating" and the resultant nanoparticle-assembled capsules as "NACs".
Selected Publications
H.G. Bagaria and M.S. Wong, "Polyamine-salt aggregate assembly of capsules as responsive drug delivery vehicles," J. Mater. Chem., 21 (26), 9454-9466 (2011). DOI:10.1039/C1JM10712G (Abstract) (Feature Article)
V.S. Murthy, S.B. Kadali, and M.S. Wong, "Polyamine-Guided Synthesis of Anisotropic, Multicompartment Microparticles", Appl. Mater. Interfac., 1, 590-596 (2009). DOI:10.1021/am8001499 (Abstract)
R.K. Rana, V.S. Murthy, J. Yu and M.S. Wong, "Nanoparticle Self-assembly of Hierarchically Ordered Microcapsule Structures", Adv. Mater. 17, 1145-1150 (2005). (Cover Article) DOI:10.1002/adma.200401612 (Abstract)