2018 Abstracts.
124. Y. Miao, N.W. Johnson, K.N. Heck, S. Guo, C.D. Powell, T Phan, D.T. Adamson, C.J. Newell, M.S. Wong, and S. Mahendra "Microbial responses to combined oxidation and catalysis treatment of 1,4-dioxane and co-contaminants in groundwater and soil" Frontiers of Environmental Science & Engineering 12 (5):2 (2018) DOI:10.1007/s11783-018-1071-6
Post-treatment impacts of a novel combined hydrogen peroxide (H2O2) oxidation and WOx/ZrO2 catalysis used for the removal of 1,4-dioxane and chlorinated volatile organic compound (CVOC) contaminants were investigated in soil and groundwater microbial community. This treatment train removed ~90% 1,4-dioxane regardless of initial concentrations of 1,4-dioxane and CVOCs. The Illumina Miseq platform and bioinformatics were used to study the changes to microbial community structure. This approach determined that dynamic shifts of microbiomes were associated with conditions specific to treatments as well as 1,4-dioxane and CVOCs mixtures. The biodiversity was observed to decrease only after oxidation under conditions that included high levels of 1,4-dioxane and CVOCs, but increased when 1,4-dioxane was present without CVOCs. WOx/ZrO2 catalysis reduced biodiversity across all conditions. Taxonomic classification demonstrated oxidative tolerance for members of the genera Massilia and Rhodococcus, while catalyst tolerance was observed for members of the genera Sphingomonas and Devosia. Linear discriminant analysis effect size was a useful statistical tool to highlight representative microbes, while the multidimensional analysis elucidated the separation of microbiomes under the low 1,4-dioxane-only condition from all other conditions containing CVOCs, as well as the differences of microbial population among original, post-oxidation, and post-catalysis states. The results of this study enhance our understanding of microbial community responses to a promising chemical treatment train, and the metagenomic analysis will help practitioners predict the microbial community status during the post-treatment period, which may have consequences for long-term management strategies that include additional biodegradation treatment or natural attenuation.
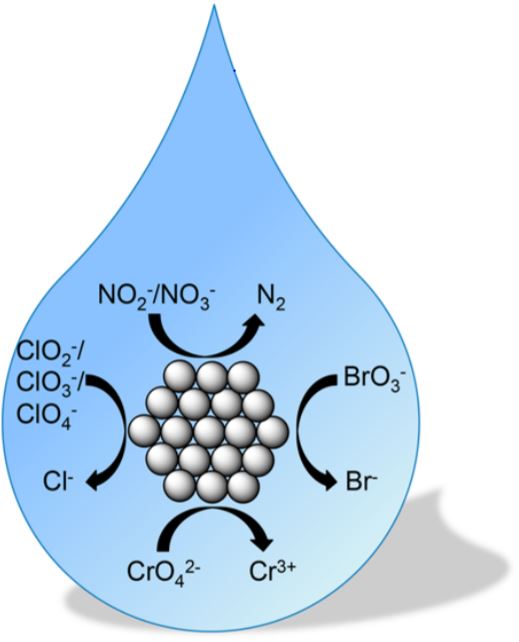
124. Y.B. Yin, S. Guo, K.N. Heck, C.A. Clark, C.L. Conrad, M.S. Wong "Treating Water by Degrading Oxyanions Using Metallic Nanostructures" ACS Sustainable Chemistry & Engineering 6 (9), pp 11160–11175 (2018) DOI:10.1021/acssuschemeng.8b02070

Consideration of the water−energy−food nexus is critical to sustainable development, as demand continues to grow along with global population growth. Cost-effective,sustainable technologies to clean water of toxic contaminants are needed. Oxyanions comprise one common class of water contaminants, with many species carrying significant human health risks. The United States Environmental Protection Agency (US EPA) regulates the concentration of oxyanion contaminants in drinking water via the National Primary Drinking Water Regulations (NPDWR). Degrading oxyanions into innocuous compounds through catalytic chemistry is a well-studied approach that does not generate additional waste, which is a significant advantage over adsorption and separation methods. Noble metal nanostructures (e.g., Au, Pd, and Pt) are particularly effective for degrading certain species, and recent literature indicates there are common features and challenges. In this Perspective, we identify the underlying principles of metal catalytic reduction chemistries, using oxyanions of nitrogen (NO2−, NO3−), chromium (CrO42−), chlorine (ClO2−, ClO3−, ClO4−), and bromine (BrO3−) as examples. We provide an assessment of practical implementation issues, and highlight additional opportunities for metal nanostructures to contribute to improved quality and sustainability of water resources.
123. P. Westerhoff, A. Atkinson, J. Fortner, M.S. Wong, J. Zimmerman, J. Gardea-Torresdey, J. Ranville, and P. Herckes "Low Risk Posed by Engineered and Incidental Nanoparticles in Drinking Water" Nature Nanotechnology 13, 661–669 (2018) DOI:10.1038/s41565-018-0217-9
Natural nanoparticles (NNPs) in rivers, lakes, oceans and ground water predate humans, but engineered nanoparticles (ENPs) are emerging as potential pollutants due to increasing regulatory and public perception concerns. This Review contrasts the sources, composition and potential occurrence of NNPs (for example, two-dimensional clays, multifunctional viruses and metal oxides) and ENPs in surface water, after centralized drinking water treatment, and in tap water. While analytical detection challenges exist, ENPs are currently orders of magnitude less common than NNPs in waters that flow into drinking water treatment plants. Because such plants are designed to remove small-sized NNPs, they are also very good at removing ENPs. Consequently, ENP concentrations in tap water are extremely low and pose low risk during ingestion. However, after leaving drinking water treatment plants, corrosion by-products released from distribution pipes or in-home premise plumbing can release incidental nanoparticles into tap water. The occurrence and toxicity of incidental nanoparticles, rather than ENPs, should therefore be the focus of future research.
122. Y. Xu, C. Ayala-Orozco, M.S. Wong, "Heavy Oil Viscosity Reduction Using Iron III para-Toluenesulfonate Hexahydrate" Society of Petroleum Engineers SPE Western Regional Meeting, 22-26 April, Garden Grove, California, USA (2018) DOI:10.2118/190020-MS
Heavy oil is a promising substitute to conventional light oil due to its abundant reserves, but its high viscosity restricts mobility and results in low recovery rate. To enhance heavy oil recovery, thermal method with metal-ligand compounds was developed, which reduced oil viscosity permanently and upgraded oil in situ. However, the mechanism for viscosity reduction using metal-ligand compounds remains unknown. This paper provides experimental observations that shed light on this mechanism. We used thermal treatment on Peace River heavy oil using iron (III) para-toluenesulfonate hexahydrate (Fe(pts)3·6H2O) at 280°C for 36 h. Thermal-only treatment changed the oil viscosity by -38.5%, while Fe(pts)3·6H2O changed by -58.5% (or nonthermal viscosity change of -20%). Gas chromatography/mass spectrometry (GC-MS) identified the formation of 4-methylbenzenethiol in oil after "thermal + Fe(pts)3·6H2O" treatment. Post-reaction analysis shown that 4-methylbenzenethiol acted as disaggregator to aid in asphaltene disaggregation. We also suggest that the metal-complex can catalytically crack certain chemical bonds in asphaltene components, contributing to further nonthermal viscosity change. This paper seeks to better understand the mechanisms of oil viscosity reduction using metal-ligand compounds. By identifying and understanding how they work, we provide new insights in developing the next generation of metal-ligand compounds.
121. K.N. Heck, S. Guo, P. Westerhoff, and M.S. Wong, "Removing Nitrates and Nitrites through New Catalysis Chemistry" Water Conditioning & Purification Magazine 3, 36-40 (2018)
Nitrate (NO3-) and nitrite (NO2-) are ubiquitous surface and groundwater contaminants, as well as a risk to human health. Existing treatment technologies such as reverse osmosis (RO) and ion exchange (IEx) separate (but do not destroy) nitrate and/or nitrite and generate brines, which require further treatment. This article discusses several catalysis approaches to ameliorate contaminated waters by directly converting (NOx-) species to non-toxic nitrogen gas and water under mild conditions.
120. S. Guo, K.N. Heck, S. Kasiraju, H. Qian, Z. Zhao, L.C. Grabow, J.T. Miller, and M.S. Wong, "Insights into Nitrate Reduction over Indium-Decorated Palladium Nanoparticle Catalysts" ACS Catalysis, 8, 503-515 (2018) DOI:10.1021/acscatal.7b01371
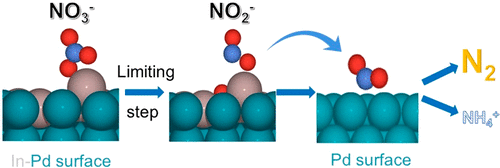
Nitrate (NO3-) is an ubiquitous groundwater contaminant and is detrimental to human health. Bimetallic palladium-based catalysts have been found to be promising for treating nitrate (and nitrite, NO2-) contaminated waters. Those containing indium (In) are unusually active, but the mechanistic explanation for catalyst performance remains largely unproven. We report that In deposited on Pd nanoparticles (NPs) (“In-on-Pd NPs”) shows room-temperature nitrate catalytic reduction activity that varies with volcano-shape dependence on In surface coverage. The most active catalyst had an In surface coverage of 40%, with a pseudo-first order normalized rate constant of kcat ∼ 7.6 L gsurface-metal-1 min-1, whereas monometallic Pd NPs and In2O3 have nondetectible activity for nitrate reduction. X-ray absorption spectroscopy (XAS) results indicated that In is in oxidized form in the as-synthesized catalyst; it reduces to zerovalent metal in the presence of H2 and reoxidizes following NO3- contact. Selectivity in excess of 95% to nontoxic N2 was observed for all the catalysts. Density functional theory (DFT) simulations suggest that submonolayer coverage amounts of metallic In provide strong binding sites for nitrate adsorption and they lower the activation barrier for the nitrate-to-nitrite reduction step. This improved understanding of the In active site expands the prospects of improved denitrification using metal-on-metal catalysts.