2016 Abstracts.
118. Y. Fang, K.N. Heck, Z. Zhao, L.A. Pretzer, N. Guo, T. Wu, J.T. Miller, and M.S. Wong, "Gold-doping of carbon-supported palladium improves reduction catalysis" Chinese Journal of Catalysia, 37(10), 1776-1786 (2016) DOI:10.1016/S1872-2067(16)62530-5
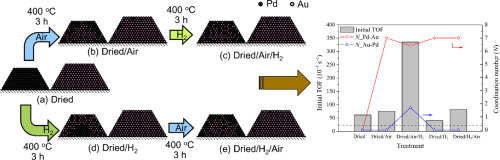
Bimetallic palladium-gold (PdAu) catalysts have better catalytic performance than monometallic catalysts for many applications. PdAu catalysts with controlled nanostructures and enhanced activities have been extensively studied but their syntheses require multiple and occasionally complicated steps. In this work, we demonstrated that supported PdAu catalysts could be simply prepared by doping a supported Pd catalyst with gold through wet impregnation and calcination. Resulting PdAu-on-carbon (PdAu/C) catalysts were tested for the room-temperature, aqueous-phase hydrodechlorination of trichloroethene. The most active PdAu/C catalyst (Pd 1.0 wt%, Au 1.1 wt%, dried/air/H2 process) had an initial turnover frequency (TOF) of 34.0 x 10-2 molTCE molPd-1 s-1, which was >15 times higher than monometallic Pd/C (Pd 1.0 wt%, initial TOF of 2.2 x 10-2 molTCE molPd-1 s-1). Through X-ray absorption spectroscopy, the gold kept Pd from oxidizing under calcination at 400°C. Probable nanostructure evolution pathways are proposed to explain the observed catalysis.
117. K.N. Heck, L.A. Pretzer, and M.S. Wong, "Nanocatalysts for Groundwater Remediation" Engineered Nanoparticles and the Environment: Biophysicochemical Processes and Toxicity, 4, 75-92 (2016)
This book chapter summarizes the current state of treating the common groundwater contaminants of organohalides and nitrates, and the opportunities for nanoengineered catalysts to treat these and emerging contaminants.
116. L. Chen, J. Zhao, S. Pradhan, B.E. Brinson, G.E. Scuseria, Z.C. Zhang, and M.S. Wong, "Ring-locking enables selective anhydrosugar synthesis from carbohydrate pyrolysis" Green Chemistry, (2016)DOI: 10.1039/C6GC01600F
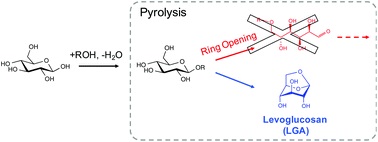
The selective production of platform chemicals from thermal conversion of biomass-derived carbohydrates is challenging. As precursors to natural products and drug molecules, anhydrosugars are difficult to synthesize from simple carbohydrates in large quantities without side products, due to various competing pathways during pyrolysis. Here we demonstrate that the nonselective chemistry of carbohydrate pyrolysis is substantially improved by alkoxy or phenoxy substitution at the anomeric carbon of glucose prior to thermal treatment. Through this ring-locking step, we found that the selectivity to 1,6-anhydro-β-D-glucopyranose (levoglucosan, LGA) increased from 2% to greater than 90% after fast pyrolysis of the resulting sugar at 600°C. DFT analysis indicated that LGA formation becomes the dominant reaction pathway when the substituent group inhibits the pyranose ring from opening and fragmenting into non-anhydrosugar products. LGA forms selectively when the activation barrier for ring-opening is significantly increased over that for 1,6-elimination, with both barriers affected by the substituent type and anomeric position. These findings introduce the ring-locking concept to sugar pyrolysis chemistry and suggest a chemical-thermal treatment approach for upgrading simple and complex carbohydrates.
115. J.B. Li, H.F. Qian, H.L. Chen, Z. Zhao, K.J. Yuan, G.X. Chen, A. Miranda, X.M. Guo, Y.J. Chen, N.F. Zheng, M.S. Wong, and J.R. Zheng, "Two distinctive energy migration pathways of monolayer molecules on metal nanoparticle surfaces" Nature Communications, 7, 10749 (2016)DOI: 10.1038/ncomms10749
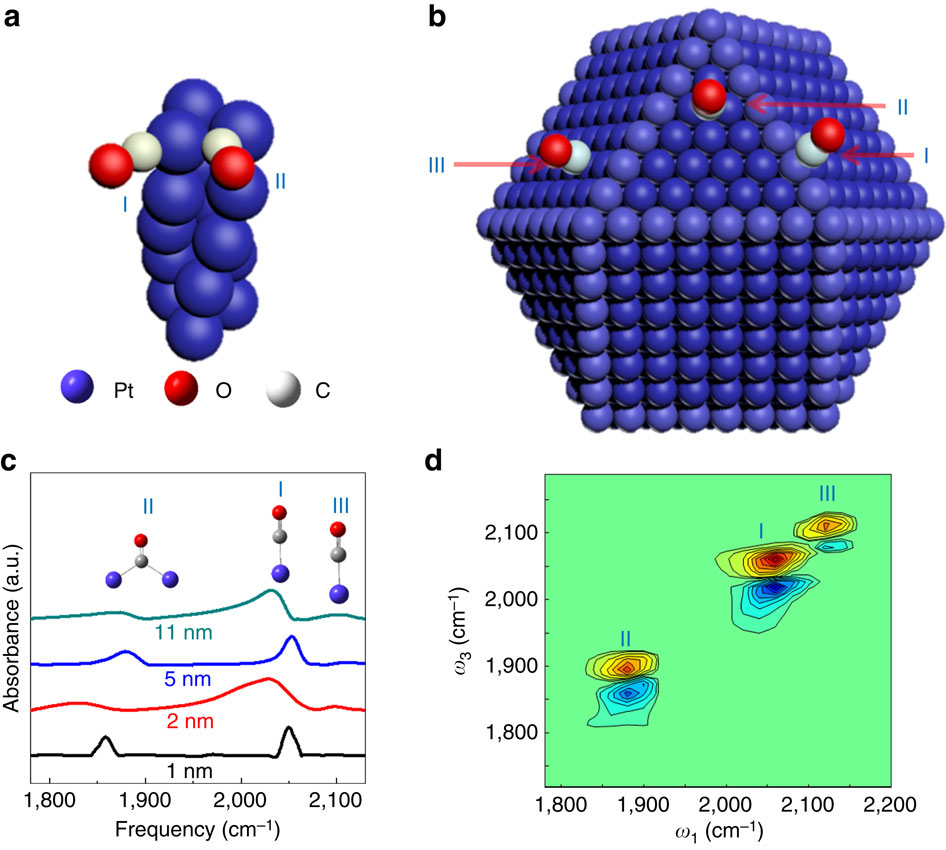
Energy migrations at metal nanomaterial surfaces are fundamentally important to heterogeneous reactions. Here we report two distinctive energy migration pathways of monolayer adsorbate molecules on differently sized metal nanoparticle surfaces investigated with ultrafast vibrational spectroscopy. On a 5-nm platinum particle, within a few picoseconds the vibrational energy of a carbon monoxide adsorbate rapidly dissipates into the particle through electron/hole pair excitations, generating heat that quickly migrates on surface. In contrast, the lack of vibration-electron coupling on approximately 1-nm particles results in vibrational energy migration among adsorbates that occurs on a twenty times slower timescale. Further investigations reveal that the rapid carbon monoxide energy relaxation is also affected by the adsorption sites and the nature of the metal but to a lesser extent. These findings reflect the dependence of electron/vibration coupling on the metallic nature, size and surface site of nanoparticles and its significance in mediating energy relaxations and migrations on nanoparticle surfaces.
114. Z. Wang, M. Gupta, S.S. Warudkar, K.R. Cox, G.J. Hirasaki, and M.S. Wong, "Improved CO2 Absorption in a Gas–Liquid Countercurrent Column Using a Ceramic Foam Contactor" Ind. Eng. Chem. Res., 55, 1387-1400 (2016) DOI: 10.1021/acs.iecr.5b03600
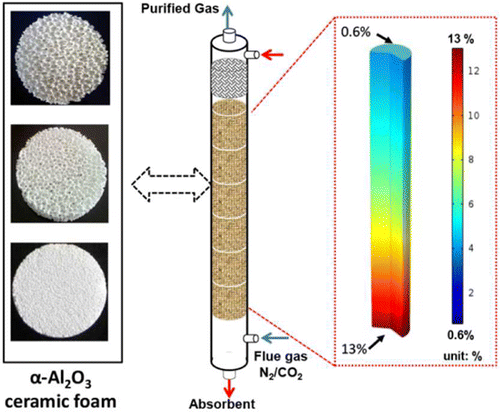
Solid foams are porous, monolithic materials with higher specific surface areas than the random packings that are commonly used in amine-based CO2 capture processes. In this work, the hydrodynamic characteristics (e.g., pressure drop, flooding point, and liquid holdup) and CO2 absorption performance of α-Al2O3 ceramic foam packings of different porosities were investigated experimentally in a gas–liquid countercurrent column. With a 30 wt % diglycolamine (DGA) solvent as the CO2 absorbent, the foams allowed higher flow rates of gas and liquid than a random packing before undesirable flooding was reached. Ceramic foams with lower porosities have larger operating capacities than those with higher porosities. A parametric study of a one-dimensional flow model was performed by investigating the effects of gas velocity, liquid velocity, and CO2 solvent loading on the CO2 removal performance. Lower gas velocities and higher liquid velocities increased the CO2 removal efficiency. The CO2 removal efficiency decreased with increasing initial CO2 loading. The initial CO2 loading of DGA solutions is recommended to be less than 0.35 mol of CO2/mol of DGA to provide efficient CO2 removal. Ceramic foams improve CO2 absorption using liquid amines, which can lead to smaller carbon capture units.
113. J.C. Velázquez, S. Leekumjorn, G.D. Hopkins, K.N. Heck, J.S. McPherson, J.A. Wilkens, B.S. Nave, M. Reinhard, and M.S. Wong, "High activity and regenerability of a palladium–gold catalyst for chloroform degradation" Journal of Chemical Technology and Biotechnology, in press (2015)DOI: 10.1002/jctb.4851
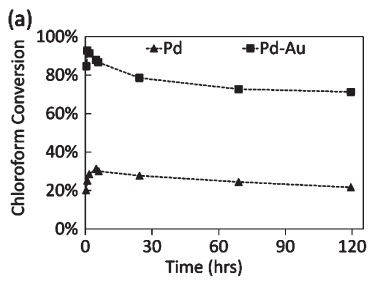
Chloroform (CF), a common groundwater contaminant, can be degraded in deionized water reductively using Pd and Pd-Au catalysts under mild conditions (room temperature, atmospheric pressure) via hydrodechlorination (HDC). However, the performance of these catalysts under field-like conditions is unknown. This study evaluates the lab-scale performance and optimal operating conditions for flow reactors using Pd/Al2O3 and Pd-Au/Al2O3.