2014 Abstracts.
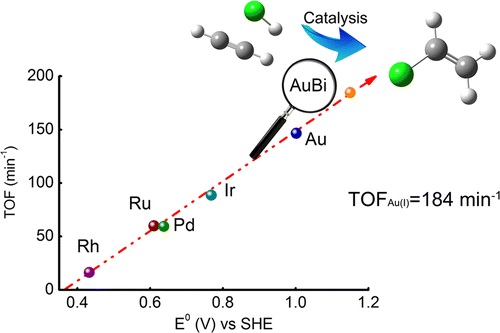
109. K. Zhou,W. Wang,Z. Zhao,G. Luo,J. T. Miller,M. S. Wong, F. Wei, "Synergistic Gold-Bismuth Catalysis for Non-Mercury Hydrochlorination of Acetylene to Vinyl-Chloride Monomer" ACS Catalysis, online (2014)DOI: 10.1021/cs500530f
Gold has been proposed as an environmentally friendly catalyst for acetylene hydrochlorination for vinyl chloride monomer synthesis by replacing the commercially used mercury catalyst. However, long life with excellent activity of is difficult to achieve since gold is readily reduced to metallic nano-particles. The stability of gold limits its industrial application. In this paper, we promoted gold with bismuth for the hydrochlorination of acetylene. It was found that the Bi promotion leads to partial reduction to AuCl, rather than the complete reduc-tion of Au to metallic nano-particles in the absence of Bi. The optimized catalyst with a molar ratio of Bi:Au=3:1 (0.3 wt% Au) showed comparable reactivi-ty to 1.0 wt% Au catalyst and significantly improved stability. Furthermore, the gold-bismuth catalyst had higher activity and stability than the commer-cial mercury catalyst, is less toxic and more envi-ronmental-friendly, making it a potentially green mercury-free industrial catalyst for acetylene hy-drochlorination.
108. V. S. Gangoli, E. H. Haroz, J. Azhang, J. Kono, T. T. Willett, R. H. Hauge, S. A. Gelwick, M. S. Wong, "Using Nonionic Surfactants for Production of Semiconductor-type Carbon Nanotubes by Gel-based Affinity Chromatography" Nanomaterials and Nanotechnology, online (2014)DOI: 10.5772/58828
Single-wall carbon nanotubes (SWCNTs) have remarkable properties based on their electronic properties, i.e., metallic or semiconducting types, but as-grown SWCNTs contain a mixture of both types. Presented here is an improved and detailed method for producing highly enriched semiconducting SWCNTs from a colloidal suspension of as-grown SWCNTs through agarose gel column-based affinity chromatography. After a 2 wt% sodium dodecyl sulphate (SDS) aqueous dispersion of SWCNTs is passed through the gel column, metal-type SWCNTs preferentially elute out using a 1.5 wt% SDS solution. Semiconductor-type SWCNTs are subsequently recovered from the column using a 2 wt% Pluronic F77 surfactant solution eluent. The semiconductor-enrichedfraction purity is in the 90-95% range, based on detailed UV-vis-NIR absorption and resonant Raman spectroscopy characterization of the particulate suspension. Semiconductor-type SWCNTs are recovered in solid form by evaporating the suspension fluid, and heating the dried sample in air to a temperature just above the Pluronic decomposition temperature. Using Pluronic and other nonionic-type surfactants can aid the scalability of the chromatographic production of semiconducting SWCNT samples.
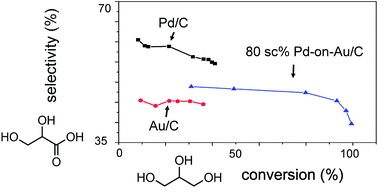
107. Z. Zhao, J. Arentz, L. A. Pretzer, P. Limpornpipat, J. M. Clomburg, R. Gonzalez, N. M. Schweitzer, T. Wu, J. T. Miller, M. S. Wong, "Volcano-shape glycerol oxidation activity of palladium-decorated gold nanoparticles" Chemical Science, online (2014)DOI: 10.1039/c4sc01001a

Bimetallic PdAu catalysts are more active than monometallic ones for the selective oxidation of alcohols, but the reasons for improvement remain insufficiently detailed. A metal-on-metal material can probe the structure–catalysis relationship more clearly than conventionally prepared bimetallics. In this study, Pdon- Au nanoparticles with variable Pd surface coverages (sc%) ranging from 10 to 300 sc% were synthesized and immobilized onto carbon (Pd-on-Au/C). Tested for glycerol oxidation at 60ºC, pH 13.5,and 1 atm under flowing oxygen, the series of Pd-on-Au/C materials showed volcano-shape catalytic activity dependence on Pd surface coverage. Increasing surface coverage led to higher catalytic activity, such that initial turnover frequency (TOF) reached a maximum of ~6000 h-1 at 80 sc%. Activity decreased above 80 sc% mostly due to catalyst deactivation. Pd-on-Au/C at 80 sc% was >10 times more active than monometallic Au/C and Pd/C, with both exhibiting TOF values less than ~500 h-1. Glyceric acid was the dominant primary reaction product for all compositions, with its zero-conversion selectivity varying monotonically as a function of Pd surface coverage. Glyceric acid yield from Pd-on-Au/C (80 sc%) was 42%, almost double the yields from Au/C and Pd/C (16% and 22%, respectively). Ex situ X-ray absorption near edge structure analysis of two Pd-on-Au/C materials with comparable activities (60 sc% and 150 sc%) showed that the former had less oxidized Pd ensembles than the latter, and that both catalysts were less oxidized compared to Pd/C. That Au stabilizes the metallic state of surface Pd atoms may be responsible for activity enhancement observed in other PdAu-catalyzed oxidation reactions. Decorating a Au surface with Pd generates a catalyst that has the deactivation resistance of Au, the higher glyceric acid selectivity of Pd, and the synergistically higher activities that neither metal has.
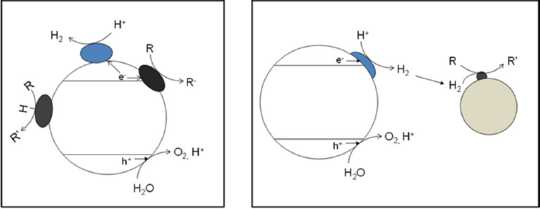
106. W. K. O’Keefe, Y. Liu, M. R. Sasges, M. S. Wong, H. Fu, T. Takata, K. Domen, "Photocatalytic Hydrodechlorination of Trace Carbon Tetrachloride (CCl4) in Aqueous Medium" Industrial & Engineering Chemistry Research, 53(23), 9600-9607 (2014)DOI: 10.1021/ie500344v
The technical feasibility of the photocatalytic hydrodechlorination of CCl4 utilizing H2 produced in situ from photocatalytic water splitting was investigated using multifunctional Pd–NiO/NaTaO3:La catalysts and a O/NaTaO3:La + Pd–Au/IX (ion-exchange resin) mixture-of-catalysts approach. In the former case, the incorporation of Pd into the water-splitting photocatalyst resulted in the reduction of H2 evolution by 3 orders of magnitude. However, the multifunctional catalyst exhibited a remarkable activity for CCl4 removal. The important catalyst parameters were elucidated by the response surface methodology. The Ni and Pd loadings and the catalyst reduction temperature had significant, nonlinear effects on the catalyst activity, indicating that both NiO and Pd nanoparticles play important roles in the photocatalytic hydrodechlorination of CCl4. The strong dependence of the turnover frequency on the catalyst parameters that govern the dispersion of the catalytic phases implies that the reaction is structure-sensitive.
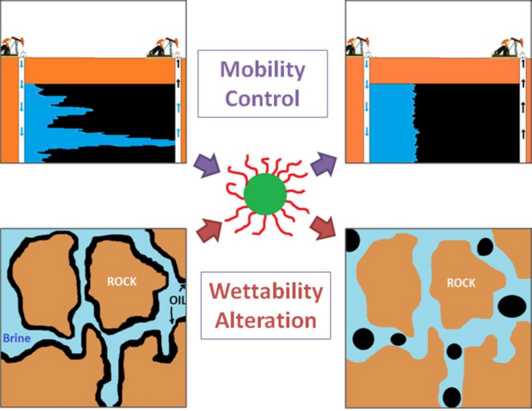
105. H. ShamsiJazeyi, C. A. Miller, M. S. Wong, J. M. Tour, R. Verduzco, "Polymer-Coated Nanoparticles for Enhanced Oil Recovery" Journal of Applied Polymer Science, in press (2014)DOI: 10.1002/app.40576
Enhanced oil recovery (EOR) processes aim to recover trapped oil left in reservoirs after primary and secondary recovery methods. New materials and additives are needed to make EOR economical in challenging reservoirs or harsh environments. Nanoparticles have been widely studied for EOR, but nanoparticles with polymer chains grafted to the surface—known as polymercoated nanoparticles (PNPs)—are an emerging class of materials that may be superior to nanoparticles for EOR due to improved solubility and stability, greater stabilization of foams and emulsions, and more facile transport through porous media. Here, we review prior research, current challenges, and future research opportunities in the application of PNPs for EOR. We focus on studies of PNPs for improving mobility control, altering surface wettability, and for investigating their transport through porous media. For each case, we highlight both fundamental studies of PNP behavior and more applied studies of their use in EOR processes. We also touch on a related class of materials comprised of surfactant and nanoparticle blends. Finally, we briefly outline the major challenges in the field, which must be addressed to successfully implement PNPs in EOR applications.
104. C. Hwang, G. Ruan, L. Wang, H. Zheng, E. L. G. Samuel, C. Xiang, W Lu, W. Kasper, K. Huang, Z. Peng, Z. Schaefer, A. T. Kan, A. A. Martí, M. S. Wong, M. B. Tomson, J. M. Tour, "Carbon-Based Nanoreporters Designed for Subsurface Hydrogen Sulfide Detection" Applied Materials & Interfaces, in press (2014)DOI: 10.1021/am5009584
Polyvinyl alcohol functionalized carbon black with H2S-sensor moieties can be pumped through oil and water in porous rock and the H2S content can be determined based on the fluorescent enhancement of the H2S-sensor addends.
103. M. C.F. Soares, M. M. Viana, Z. L. Schaefer, V. S. Gangoli, Y. Cheng, V. Caliman, M. S. Wong, G. G. Silva, "Surface modification of carbon black nanoparticles by dodecylamine: thermal stability and phase transfer in brine medium" Carbon, in press (2014)DOI: 10.1016/j.carbon.2014.02.008
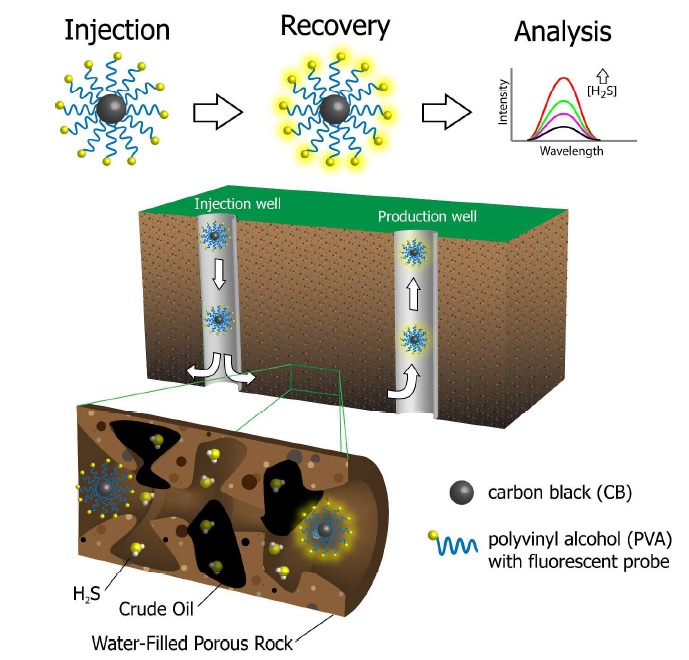
Stable aqueous dispersions of carbon black (CB) nanoparticles were prepared by developing a generic bilayer approach to the CB phase-transfer method using commercial Avanel as a surfactant. CB was oxidized using ammonium persulfate, and dodecylamine (DDA) was grafted onto this carbon core using N,N-dicyclohexylcarbodiimide as a coupling agent. The covalent bond between CB and DDA was confirmed by X-ray photoelectron spectroscopy, Fourier transform infrared spectroscopy, ultraviolet–visible spectroscopy, and thermogravimetric analysis. The average diameter of the primary carbon nanoparticles after chemical modification increased from 15 to 20 nm, as determined by high-resolution transmission electron microscopy. Dispersions of the modified CB was stable in organic solvents and were thermally stable in saline water when Avanel was used as a surfactant. The Avanel coating on the CB particles enabled the design of carbon materials with high colloidal stability, as evidenced by dynamic light scattering measurements and the breakthrough characteristics of the coated material in sandstone rocks.
102. G. C. Kini, J. Yu, L. Wang, A. T. Kan, S. L. Biswal, J. M. Tour, M. B. Tomson, M. S. Wong, "Salt- and temperature-stable quantum dot nanoparticles for porous media flow" Colloids Surf.A: Physicochem. Eng. Aspects, 443, 492-500 (2014)DOI: 10.1016/j.colsurfa.2013.11.042
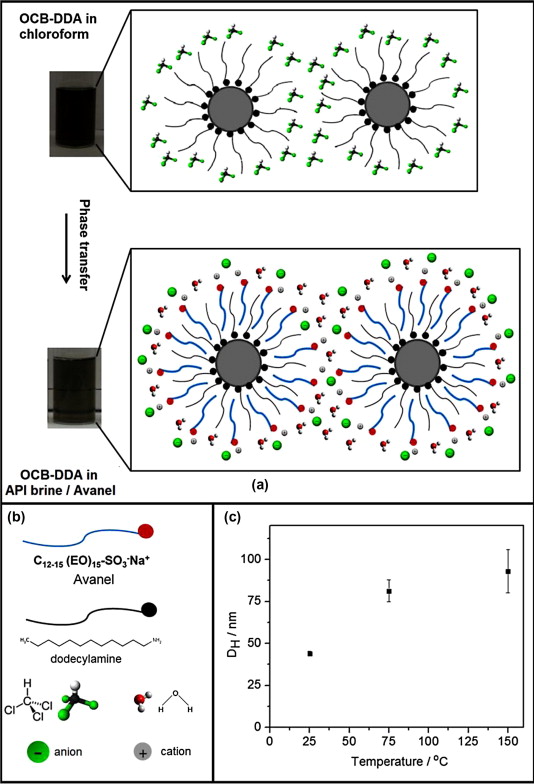
The transport of colloidal nanoparticles (NPs) through porous media is a well-studied phenomenon at ambient temperature and in low-to-zero salinity water found in aquatic systems. Little is known at much higher temperatures and salinities such as conditions found in petroleum reservoirs, thus limiting the possible use of NPs in downhole oilfield applications. Using 3-nm CdSe quantum dots (QDs) as a model material, we report that NPs can be prepared with excellent colloidal stability at high ionic strengths and elevated temperatures. QDs with an outer coating of a nonionic ethoxylated alcohol surfactant showed little aggregation in synthetic seawater with ionic strength of 0.55 M and 1 M NaCl brine solution based on dynamic light scattering analysis. They showed colloidal stability up to 70°C, close to the cloud point temperature of the nonionic surfactant. They further showed nearly unimpeded flow behavior when carried in high-salinity water through a packed column of crushed calcite or sandstone mineral at room temperature. QDs were successfully passed through a medium-permeability Berea sandstone core (100 mDa) at 17 atm, 25 °C, and 8 mL/h. The preparation strategy for salt- and temperature-stable QDs is applicable to a wide range of particle sizes and compositions, toward the general handling and use of functional NPs in high-salinity environments.
101. H. F. Qian, Z. Zhao, J. C. Velazquez, L. A. Pretzer, K. N. Hecka, M. S. Wong, "Supporting palladium metal on gold nanoparticles improves its catalysis for nitrite reduction" Nanoscale, 59(12), 4474–4482 (2013). DOI: 10.1039/C3NR04540D
Nitrate (NO3-) and nitrite (NO2-) anions are often found in groundwater and surface water as contaminants globally, especially in agricultural areas due to nitrate-rich fertilizer use. One popular approach to studying the removal of nitrite/nitrate from water has been their degradation to dinitrogen via Pd-based reduction catalysis. However, little progress has been made towards understanding how the catalyst structure can improve activity. Focusing on the catalytic reduction of nitrite in this study, we report that Au NPs supporting Pd metal ("Pd-on-Au NPs") show catalytic activity that varies with volcano-shape dependence on Pd surface coverage. At room temperature, in CO2- buffered water, and under H2 headspace, the NPs were maximally active at a Pd surface coverage of 80%, with a first-order rate constant (kcat = 576 L gPd-1 min-1) that was 15x and 7.5x higher than monometallic Pd NPs (~4 nm; 40 L gPd-1 min-1) and Pd/Al2O3 (1 wt% Pd; 76 L gPd-1 min-1), respectively. Accounting only for surface Pd atoms, these NPs (576 L gsurface-Pd-1 min-1) were 3.6x and 1.6x higher than monometallic Pd NPs (160 L gsurface-Pd-1 min-1) and Pd/Al2O3 (361 L gsurface-Pd-1 min-1). These NPs retained ~98% of catalytic activity at a chloride concentration of 1 mM, whereas Pd/Al2O3 lost ~50%. The Pd-on-Au nanostructure is a promising approach to improve the catalytic reduction process for nitrite and, with further development, also for nitrate anions.