2012 Abstracts.
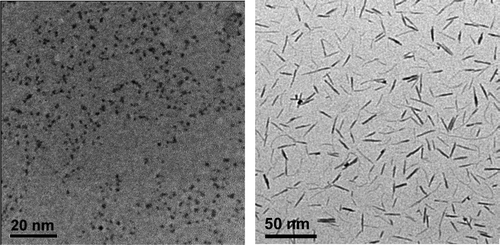
91. N. Soultanidis, W. Zhou, C. J. Kiely, and M. S. Wong, "Solvothermal Synthesis of ultrasmall tungsten oxide nanoparticles," Langmuir, 28 (51), 17771–17777 (2012). DOI: 10.1021/la3029462
The synthesis of catalytically useful, ultrasmall oxide nanoparticles (NPs) of group 5 and 6 metals is not readily achievable through reported methods. In this work, we introduce a one-pot, two-precursor synthesis route to 2 nm MOx NPs in which a polyoxometalate salt is decomposed thermally in a high-boiling organic solvent oleylamine. The use of ammonium metatungstate resulted in oleylamine-coated, crystalline WOx NPs at consistently high yields of 92 ± 5%. The semicrystalline NPs contained 20–36 WOx structural units per particle, as determined from aberration-corrected high-resolution scanning transmission electron microscopy, and an organic coating of 16–20 oleylamine molecules, as determined by thermogravimetric analysis. The NPs had a mean size of 1.6 ± 0.3 nm, as estimated from atomic force microscopy and small-angle X-ray scattering measurements. Carrying out the synthesis in the presence of organic oxidant trimethylamine N-oxide led to smaller WOx NPs (1.0 ± 0.4 nm), whereas the reductant 1,12-dodecanediol led to WOx nanorods (4 ± 1 nm × 20 ± 5 nm). These findings provide a new method to control the size and shape of transition metal oxide NPs, which will be especially useful in catalysis.
90. S. Gullapalli, J. M. Grider, H. G. Bagaria, K.-S. Lee, M. Cho, V. L. Colvin, G. E. Jabbour, and M. S. Wong, "Molten-droplet synthesis of composite CdSe hollow nanoparticles," Nanotechnology, 23 (49), 495605-495615 (2012). DOI: 10.1088/0957-4484/23/49/495605
Many colloidal synthesis routes are not scalable to high production rates, especially for nanoparticles of complex shape or composition, due to precursor expense and hazards, low yields, and the large number of processing steps. The present work describes a strategy to synthesize hollow nanoparticles (HNPs) out of metal chalcogenides, based on the slow heating of a low-melting-point metal salt, an elemental chalcogen, and an alkylammonium surfactant in octadecene solvent. The synthesis and characterization of CdSe HNPs with an outer diameter of 15.6 ± 3.5 nm and a shell thickness of 5.4 ± 0.9 nm are specifically detailed here. The HNP synthesis is proposed to proceed with the formation of alkylammonium-stabilized nano-sized droplets of molten cadmium salt, which then come into contact with dissolved selenium species to form a CdSe shell at the droplet surface. In a reaction–diffusion mechanism similar to the nanoscale Kirkendall effect it is speculated that the cadmium migrates outwardly through this shell to react with more selenium, causing the CdSe shell to thicken. The proposed CdSe HNP structure comprises a polycrystalline CdSe shell coated with a thin layer of amorphous selenium. Photovoltaic device characterization indicates that HNPs have improved electron transport characteristics compared to standard CdSe quantum dots, possibly due to this selenium layer. The HNPs are colloidally stable in organic solvents even though carboxylate, phosphine, and amine ligands are absent; stability is attributed to octadecene-selenide species bound to the particle surface. This scalable synthesis method presents opportunities to generate hollow nanoparticles with increased structural and compositional variety.
89. M. Thakur, S. L. Sinsabaugh, M. J. Isaacson, M. S. Wong, and S. L. Biswal, "Inexpensive method for producing macroporous silicon particulates (MPSPs) with pyrolyzed polyacrylonitrile for lithium ion batteries," Scientific Reports, 2: 795, 1-7 (2012). DOI: 10.1038/srep00795
One of the most exciting areas in lithium ion batteries is engineering structured silicon anodes. These new materials promise to lead the next generation of batteries with significantly higher reversible charge capacity than current technologies. One drawback of these materials is that their production involves costly processing steps, limiting their application in commercial lithium ion batteries. In this report we present an inexpensive method for synthesizing macroporous silicon particulates (MPSPs). After being mixed with polyacrylonitrile (PAN) and pyrolyzed, MPSPs can alloy with lithium, resulting in capacities of 1000 mAhg-1 for over 600+ cycles. These sponge-like MPSPs with pyrolyzed PAN (PPAN) can accommodate the large volume expansion associated with silicon lithiation. This performance combined with low cost processing yields a competitive anode material that will have an immediate and direct application in lithium ion batteries.
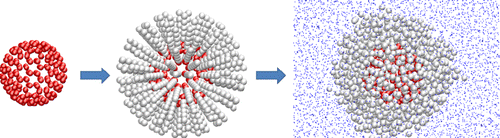
88. S. Leekumjorn, S. Gullapalli, and M. S. Wong, "Understanding the solvent polarity effects on surfactant-capped nanoparticles," J. Phys. Chem. B, 116 (43), 13063–13070 (2012). DOI: 10.1021/jp307985c

Understanding the molecular interactions between suspended nanoparticles (NPs) and the suspending solvent fluid may provide a useful avenue to create and to study exotic NP ensembles. This study focused on using a coarse-grained computational model to investigate the molecular interactions between oleate-capped NPs in various solvents, and to relate the results to experimental features of solvent-suspended, oleate-capped CdSe quantum dots (QDs). The QDs were modeled as a closed-shell fullerene molecule with an oleate-like ligand attached to each vertex. Solvent polarity was found to correlate to the simulation and experimental results more strongly than either dielectric constant or dipole moment. Computational results showed that the nonpolar solvents of hexane, toluene, and benzene (polarity index ETN 0.120) kept NPs in suspension and solvated the oleate chains such that the oleate layer swelled to full extension. In contrast, as the most polar solvent tested (ETN = 1.000), water caused NPs to aggregate and precipitate. It partially solvated the oleate chains and compressed the layer to 86% of full extension. For solvents of intermediate polarity like ethanol, acetone, and chloroform, the oleate layer swelled with decreasing polarity index values, with rapid swelling occurring close to ETN = 0.307 (50:50 vol % chloroform/acetone) below which QDs were colloidally stable. This study represents the first attempt to delineate the solvent effect on surfactant-coated NP hydrodynamic size, colloidal stability, and aggregation behavior.
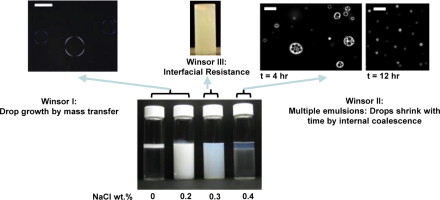
87. G. C. Kini, S. L. Biswal, M. S. Wong, and C. A. Miller, "Characteristics of spontaneously formed nanoemulsions in octane/AOT/brine systems," J. Colloid Interface Sci., 385 (1), 111-121 (2012). DOI: 10.1016/j.jcis.2012.07.041
Nanoemulsions were formed spontaneously by diluting water-in-oil (W/O) or brine-in-oil (B/O) microemulsions of a hydrocarbon (octane), anionic surfactant (Aerosol-OT or AOT) and water or NaCl brine in varying levels of excess brine. The water-continuous nanoemulsions were characterized by interfacial tension, dynamic light scattering, electrophoresis, optical microscopy and phase-behavior studies. The mechanism of emulsification was local supersaturation and resulting nucleation of oil during inversion. For nanoemulsions formed at low salinities with Winsor I phase behavior, octane drops grew from initial diameters of 150–250 nm to 480–1000 nm over 24 h, depending on salinity. Growth was caused by mass transfer but seemed to approach the asymptotic stage of Ostwald ripening described by the Lifshitz–Slyozov–Wagner (LSW) theory only for dilution with salt-free water. Near the higher cross-over salinity (Winsor III), the nanoemulsions showed much slower growth with droplet size consistently remaining below 200 nm over 24 h and reaching 250 nm after 1 week. Birefringence indicated the presence of liquid crystal for these conditions, which could have contributed to the slow growth rate. At even higher salinity levels in the Winsor II domain, W/O/W multiple emulsions having drops greater than 1 nm in diameter were consistently recorded for the first 5–7 h, after which size decreased to values below 1 nm. The number and size of internal water droplets in multiple emulsion drops was found to decrease over time, suggesting coalescence of internal droplets with the continuous water phase and mass transfer of water from internal droplets to continuous phase as possible mechanisms of the observed drop shrinkage. Electrophoresis studies showed the nanoemulsions to be highly negatively charged (zeta potentials of ?60 mV to ?120 mV). The high charge on octane droplets helped assure stability to flocculation and coalescence, thereby allowing mass transfer to control growth in the Winsor I and III regions.
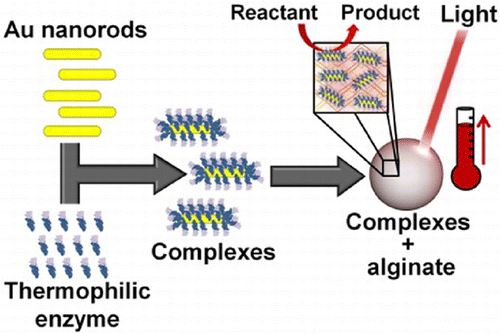
86. M. Thakur, R. B. Pernites, N. Nitta, M. Isaacson, S. L. Sinsabaugh, M. S. Wong, and S. L. Biswal, "Freestanding Macroporous Silicon and Pyrolyzed Polyacrylonitrile As a Composite Anode for Lithium Ion Batteries," Chem. Mater., 24 (15), 2998–3003 (2012). DOI: 10.1021/cm301376t
Silicon continues to draw great interest as an anode material for lithium ion batteries due to its large specific capacity for lithium. Macroporous silicon produced by electrochemical etching is one of several anode materials of interest, but its energy density is oftentimes limited due to its attachment to an unreactive silicon substrate. Here, we present a novel “lift-off” method by which a freestanding macroporous silicon film (MPSF) is electrochemically detached from the underlying bulk silicon and combined with pyrolyzed polyacrylonitrile (PAN), a conductive polymer that forms a conjugated-chain chemical structure. We report the performance of these silicon thin films with and without pyrolyzed PAN.
85. K. -P. Chao, H. Bagaria, M. S. Wong, and S. L. Biswal, "Templating CdSe tetrapods at the air/water interface with POPC lipids," J. Colloid Interface Sci., 378 (1), 58-63 (2012). DOI:10.1016/j.jcis.2012.04.028

Surfactants have been widely used as templating agents to pattern the orientation of nanoparticles of various conformations. Here we report the use of a lipid, 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine (POPC), as a template to order CdSe tetrapods (TPs) at the air/water interface using a Langmuir–Blodgett trough. The surface pressure versus area isotherms for CdSe TPs and CdSe TPs/POPC are examined and monitored by Brewster angle microscopy (BAM). The transferred thin films are further characterized by transmission electron microscopy (TEM) and atomic force microscopy (AFM). Initially disc-like structures containing randomly oriented TPs form during solvent evaporation. Upon decreasing surface area, these discs merge into larger continental structures. In a mixed CdSe TPs/POPC system, these discs organize into wire-like networks upon compression. We detail how lipid molecules can be used to direct the two-dimensional assembly of TPs.
84. C. -C. Hwang, L. Wang, W. Lu, G. -D. Ruan, G. C. Kini, C. -S. Xiang, E. L. G. Samuel, W. Shi, A. T. Kan, M. S. Wong, M. B. Tomson, and J. M. Tour, "Highly stable carbon nanoparticles designed for downhole hydrocarbon detection," Energy Environ. Sci., 5, 8304-8309 (2012). DOI:10.1039/C2EE21574H
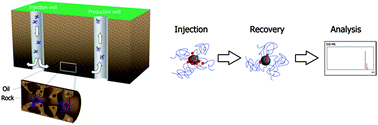
Sulfated polyvinyl alcohol functionalized carbon black, stable under high temperature and high salinity conditions, efficiently carries a hydrophobic compound through a variety of oil-field rock types and releases the compound when the rock contains hydrocarbons.
83. S. -J. Li, Y.-L. Fang, C. D. Romanczuk, Z.-H. Jin, T.-L, Li, and M. S. Wong, "Establishing the Trichloroethene Dechlorination Rates of Palladium-based Catalysts and Iron-based Reductants," Appl. Catal. B-Environ., 125, 95-102 (2012). DOI:10.1016/j.apcatb.2012.05.025
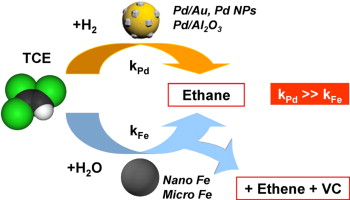
The removal of undesired chlorinated hydrocarbon contaminants through chemical destruction using ex situ Pd-based catalytic or in situ Fe-based reductive nanomaterials offers unique advantages over current physical displacement methods for groundwater treatment. While these two types of chemical methods has been studied in-depth in recent years, their respective hydrodechlorination and dechlorination transformations have not been analyzed together before. Here, the reactivities of Pd catalysts and Fe reductants were experimentally assessed for trichloroethene (TCE) degradation using room-temperature, atmospheric-pressure, dihydrogen-headspace-filled batch reactor studies under buffered and non-buffered conditions. Pseudo-first order reaction rate constants at pH 7 spanned 9 decades: 1.2 × 104, 1.0 × 103, 4.5 × 102, 2.41 × 10?4, 4.2 × 10?4, and 7.09 × 10?6 L/gactive-metal/min for Pd-on-Au nanoparticles (Pd/Au NPs), Pd NPs, Pd-on-alumina powder, and two nano-sized forms and one micron-sized form of commercial zerovalent iron, respectively. With rates measured in the range of commonly reported values, the Fe-based materials produced ethane, ethene, and vinyl chloride; ethene hydrogenated into ethane at sufficiently long reaction times. The much more active Pd-based materials produced ethane as the primary TCE degradation reaction product. This study presents, for the first time, a quantitative comparison of TCE degradation rates determined under identical experimental conditions.
82. O. Kuznetsov, Y.-Q. Sun, R. Thaner, A. Bratt, V. Shenoy, M. S. Wong, J. Jones, and W. E. Billups, "Water-Soluble Nanodiamond," Langmuir, 28 (11), 5243–5248 (2012). DOI:10.1021/la204660h

Reduction of the graphenic edges of annealed nanodiamond by sodium in liquid ammonia leads to a nanodiamond salt that reacts with either alkyl or aryl halides by electron transfer to yield radical anions that dissociate spontaneously into free radicals and halide. The free radicals were observed to add readily to the aromatic rings of the annealed nanodiamond. Nanodiamonds functionalized by phenyl radicals were sulfonated in oleum, and the resulting sulfonic acid was converted to the sodium salt by treatment with sodium hydroxide. The solubility of the salt in water was determined to be 248 mg/L. Nanodiamond functionalized by carboxylic acid groups could be prepared by reacting 5-bromovaleric acid with the annealed nanodiamond salt. The solubility of the sodium carboxylate in water was found to be 160 mg/L.
81. Q. X. Nguyen, T. G. Belgard, J. J. Taylor, V. S. Murthy, N. J. Halas, and M. S. Wong, "Water-Phase Synthesis of Cationic Silica/Polyamine Nanoparticles," Chem. Mater. 24 (8), 1426–1433 (2012) DOI:10.1021/cm203132m

Silica particles are commonly functionalized with amine groups on their surface through the hydrolytic condensation of aminotrialkoxysilanes for use in bioimaging, enzyme immobilization, and other applications. Eliminating this aminotrialkoxysilane condensation step could simplify and improve the efficiency of the synthesis of amine-functionalized silica. Here, we describe a one-pot, ambient-condition, water-phase method to synthesize silica-based nanoparticles (NPs) that present surface amine groups. The formation mechanism involves the electrostatic cross-linking of cationic polyallylamine hydrochloride by citrate anions and the infusion of the resulting polymer/salt aggregates by silicic acid. The particles were unimodal with average diameters in the range of 40 to 100 nm, as determined by the size of the templating polymer-salt aggregates. Colorimetric analysis using Coomassie brilliant blue and zeta potential measurements confirmed the presence of surface amine groups on the hybrid silica/polymer NPs. The point of zero-charge value for these NPs was 5, between the corresponding values of unfunctionalized and aminopropyltriethoxysilane-functionalized silica particles (~2 and ~10, respectively). Surface charge calculations indicated the hybrid NPs had a lower amine surface density than aminopropyltriethoxysilane-functionalized silica (0.057 #/nm2 vs 0.169 #/nm2 at pH 7). The polymer-salt aggregate synthesis chemistry is a new approach toward controlling the amine surface density and point of zero-charge of hybrid silica/polymer NPs.
80. L. Y. Zhu, W.-J. An, J. W. Springer, L. B. Modesto-Lopez, S. Gullapalli, D. Holten, M. S. Wong and P. Biswas, "Linker-free quantum dot sensitized TiO2 photoelectrochemical cells," Int. J. Hydrogen Energy, 37 (8), 6422–6430 (2012). DOI:10.1016/j.ijhydene.2012.01.028
Aerosol based techniques were used to characterize and deposit quantum dots (QDs). Using an electrospray-assisted characterization technique, the mobility diameter of CdSe QDs was successfully measured in real-time. The electrospray technique was also used to deposit CdSe QDs onto nanostructured TiO2 films. Compared to conventional methodologies such as dip coating with linker-containing molecules or chemical bath deposition, an electrospray system enabled uniform deposition of QDs over the nanostructured TiO2 surface in a short processing time. As-deposited films were annealed to enhance binding between the QDs and TiO2 surface. These QD-decorated TiO2 films were used in photoelectrochemical cells, for which the photoenergy conversion efficiencies were tested. Optimization of the deposition time of the QDs resulted in increased efficiencies. Multiple layers of QDs caused a decrease in energy-conversion efficiency, likely due to inhibition of the transportation of photogenerated electrons into the TiO2 structure. The energy-conversion efficiency trends were supported by time-resolved photoluminescence decay data.
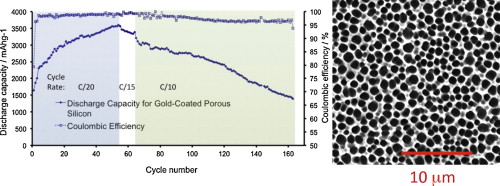
78. M. Thakur, M. Isaacson, S. L. Sinsabaugh, M. S. Wong, and S. L. Biswal, "Gold-Coated porous silicon films as anodes for lithium ion batteries," J. Power. Sources., 205, 426-432 (2012). DOI:10.1016/j/jpowsour.2012.01.058
Silicon has the highest known theoretical charge capacity for lithium, making it a promising material for rechargeable lithium ion batteries (LIB). A significant drawback of silicon anodes is the large volume change associated with the insertion and extraction of lithium, which oftentimes leads to cracking and pulverization of the anode, limiting its cycle life. We present a layered architecture consisting of a gold-coated porous silicon film attached to a bulk silicon substrate. This structure demonstrates an enhanced ability to alloy with lithium ions over several charge/discharge cycles while maintaining mechanical integrity. With this structure we show that a specific capacity of over 3000 mAh g-1 can be achieved for over 50 charge–discharge cycles at 100 ?A cm-2, and 2500 mAh g-1 can be achieved for over 75 cycles with coulombic efficiencies over 95%. This is a significant improvement over a gold-coated, non-porous silicon sample, which had a maximum capacity of 1 mAh g-1 before failing after 10 cycles at 0.25 mAh g-1 when cycled at a constant current of 100 ?A cm-2, illustrating the benefit of internal pores. Gold-coated porous silicon out-performed non-gold-coated porous silicon, which had a first cycle discharge capacities of 500 mAh g-1, which quickly faded to 76 mAh g-1 after the 10th cycle when cycled at a constant current of 50 ?A cm-2. The combination of internal pores and a gold-coating points to a new approach to improving the long-term cycleability and high-capacity performance metrics of LIB anodes.