2008 Abstracts.
48. Q. Li, M. B. Tomson, M. S. Wong, and P. J. J. Alvarez, "Nanotechnology: A Source for Answers to the Global Clean Water Challenge" Water 21, Magazine of the International Water Association, August 26-28 (2008)
No Abstract.
47. M.A. Yaseen, J. Yu, M.S. Wong and B. Anvari, "In-vivo Fluorescence Imaging of Mammalian Organs using Charge-assembled Mesocapsule Constructs Containing Indocyanine Green" Optics Express, 16 (25), 20577-20587 (2008). DOI:10.1364/OE.16.020577
Indocyanine green (ICG) is a fluorescent probe used in clinical imaging. However, its utility remains limited by optical instability, rapid circulation kinetics, and exclusive uptake by the liver. Using mesocapsule (MC) constructs to encapsulate ICG, we have developed a technology to stabilize ICG’s optical properties and alter its biodistribution. We present in vivo fluorescence images of mammalian organs to demonstrate the potential application of our ICG encapsulation technology for optical imaging of specific tissues.
46. K.N. Heck, B.G. Janesko, G.E. Scuseria, N.J. Halas and M.S.Wong, "Observing Metal-Catalyzed Chemical Reactions in Situ Using Surface-Enhanced Raman Spectroscopy on Pd-Au Nanoshells" J. Am. Chem. Soc. , 130 (49), 16592–16600 (2008). DOI:10.1021/ja803556k
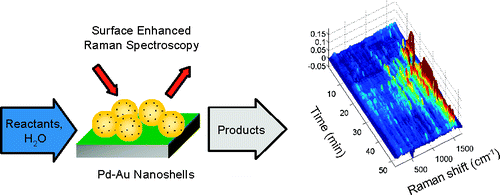
Insight into the nature of transient reaction intermediates and mechanistic pathways involved in heterogeneously catalyzed chemical reactions is obtainable from a number of surface spectroscopic techniques. Carrying out these investigations under actual reaction conditions is preferred but remains challenging, especially for catalytic reactions that occur in water. Here, we report the direct spectroscopic study of the catalytic hydrodechlorination of 1,1-dichloroethene in H2O using surface-enhanced Raman spectroscopy (SERS). With Pd islands grown on Au nanoshell films, this reaction can be followed in situ using SERS, exploiting the high enhancements and large active area of Au nanoshell SERS substrates, the transparency of Raman spectroscopy to aqueous solvents, and the catalytic activity enhancement of Pd by the underlying Au metal. The formation and subsequent transformation of several adsorbate species was observed. These results provide the first direct evidence of the room-temperature catalytic hydrodechlorination of a chlorinated solvent, a potentially important pathway for groundwater cleanup, as a sequence of dechlorination and hydrogenation steps. More broadly, the results highlight the exciting prospects of studying catalytic processes in water in situ, like those involved in biomass conversion and proton-exchange membrane fuel cells.
45. M. S. Wong, Book review of Nanoparticles and Catalysis D. Astruc, Ed.; Angew. Chem. 47, 7795-7796 (2008). DOI:10.1002/anie.200785570
No abstract.
44. V. S. Murthy and M. S. Wong, "Enzyme Encapsulation using Nanoparticle-assembled Capsules" in Nanoscale Science and Technology in Biomolecular Catalysis; J. B. Kim, S. H. Kim, and P. Wang, Eds., American Chemical Society: Washington, D.C.; Chapter 13, 214-232 (2008) DOI:10.1021/bk-2008-0986.ch013
We discuss the synthesis and characterization of enzyme-containing nanoparticle-assembled capsules (NACs) using acid phosphatase as the model enzyme. NACs are a new capsular material that can be synthesized rapdily under ambient conditions and mild pH values. Unlike vesiclesand other organic-based hollow sphere structures, NACs can be handled in the absence of water. The hollow forms spontaneously, allowing for non-destructive encapsulation. The enzyme is shown to preserve its activity within the capsules, and a maximum loading and encapsulation efficiency of ~18 wt% and ~70%, respectively, can be achieved. The encapsulated enzymes can be recovered and reused through centrifugation.
43. E. I. Ross-Medgaarden, W. V. Knowles, T. Kim, M. S. Wong, W. Zhou, C. J. Kiely, and I. E. Wachs, "New Insights into the Nature of the Acidic Catalytic Active Sites Present in ZrO2-supported Tungsten Oxide Catalysts" J. Catal. 256, 108-125 (2008) DOI:10.1016/j.jcat.2008.03.003
An extensive series of supported WO3/ZrOx(OH)4-2x catalysts (WZrOH) were synthesized by standard aqueous impregnation of ammonium metatungstate into an amorphous ZrOx(OH)4-2x metastable support, followed by high-temperature calcination (at 773–1173 K). The supported WZrOH catalysts were also compared with well-defined model supported WO3/ZrO2 catalysts (WZrO2) consisting of a thermally stable crystalline m-ZrO2 support. Both series of supported tungsten oxide catalysts were physically characterized (by XRD, XPS, TEM, in situ Raman, and in situ UV–vis spectroscopy) and chemically probed by methanol dehydration (i.e., TPSR spectroscopy and steady-state catalytic studies). Monolayer surface WOx coverage was found to occur at ~4.5–5 W-atoms/nm2 for both catalytic systems. Whereas the dehydrated model supported WZrO2 series contained only surface WOx species below monolayer coverage, the dehydrated supported WZrOH series had the same surface WOx species, as well as some Zr-stabilized distorted WO3 nanoparticles (NPs). Above monolayer coverage, the model supported WZrO2 catalysts contained only ordered crystalline WO3 NPs, but the supported WZrOH catalysts had both ordered WO3 NPs and Zr-stabilized distorted WO3 NPs. The comparative methanol dehydration to dimethyl ether acidity study revealed that the Zr-stabilized distorted WO3 NPs were the catalytic active sites in supported WZrOH catalysts. These findings represent a new model for the origin of the enhanced solid acidity of supported WZrOH catalysts.
42. S. B. Kadali, N. Soultanidis, and M. S. Wong, "Assembling Colloidal Silica into Porous Hollow Microspheres" Topic. Catal., 49 (3-4), 251-258 (2008) DOI:10.1007/s11244-008-9079-y
A non-surfactant-based synthesis approach to mesoporous hollow spheres through the use of colloidal silica is presented. Based on nanoparticle assembly chemistry developed previously for silica/polymer hybrid microcapsules, the room-temperature preparation follows a two-step sequence: (1) the electrostatic reaction of cationic polymer with an anionic salt solution, resulting in a suspension of salt-bridged polymer aggregates; and (2) the electrostatic reaction between this suspension and an aqueous suspension of nanoparticles (NPs). As a specific example, 13-nm silica particles, combined with polyallylamine and sodium citrate, gave silica/polymer hollow spheres with a mean diameter of 2.1 m and a BET surface area of 4 m2/g. After calcination at 600°C, the resulting silica-only microcapsules had a BET surface area of 259 m2/g,