2001 Abstracts.
6. M. S. Wong, E. S. Jeng and J. Y. Ying, "Supramolecular Templating of Thermally Stable Crystalline Mesoporous Metal Oxides Using Nanoparticulate Precursors" Nano Lett. 1, 637-642 (2001). DOI:10.1021/nl015594y
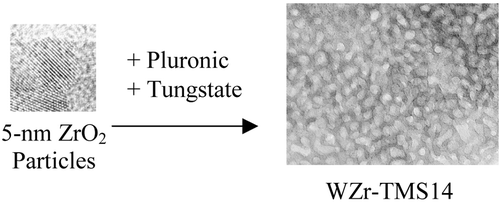
A supramolecular templating approach to the preparation of crystalline mesoporous metal oxides using inorganic colloidal nanoparticles as building blocks is presented. The nanoparticles are used to form a mesostructured material with a triblock copolymer surfactant as the templating agent and with a tungstate salt as the “binding” agent. Tungstated zirconium oxide produced in this manner was found to be highly thermally stable (in excess of 600°C) while maintaining mesoporosity and a high surface area. Other mesoporous oxides were produced using zirconium salts, titania nanoparticles, and aluminum polycations, illustrating the flexibility of this “nanoparticle/surfactant templating” route.
5. X. Gao, I. E. Wachs, M. S. Wong and J. Y. Ying, "Structural and Reactivity Properties of Nb-MCM-41: Comparison with That of Highly Dispersed Nb2O5/SiO2 Catalysts" J. Catal. 203, 18-24 (2001). DOI:10.1006/jcat.2001.3313
The molecular structures of Nb cations in mesoporous material Nb---MCM-41 and the supported Nb2O5/SiO2 catalyst under hydrated and dehydrated conditions were investigated by in-situ Raman and UV-vis-NIR diffuse reflectance spectroscopies. Methanol oxidation was employed as a chemical probe reaction to examine reactivity/selectivity properties of these catalytic materials. The structural characterization techniques demonstrate that similar surface Nb oxide species are present in both Nb---MCM-41 and Nb2O5/SiO2 catalysts, which are sensitive to the environmental conditions (ambient or dehydrated). However, the characterization results also suggest that some Nb atoms in Nb---MCM-41 may be incorporated within the siliceous framework. The spectroscopic results revealed that under dehydrated conditions, the Nb cations in Nb---MCM-41 and 1% Nb2O5/SiO2 are predominantly composed of isolated NbO4 units, while the Nb cations in the supported Nb2O5/SiO2 catalysts possess polymerized NbOx species and/or bulk Nb2O5 with increasing Nb loading. The catalytic results indicate that the dispersed Nb cations in both types of catalysts are active redox sites for methanol oxidation and exhibit similar reactivity and selectivity properties due to the presence of Nb---O---Si bonds in their structures.