Biomass Upgrading Chemistry.
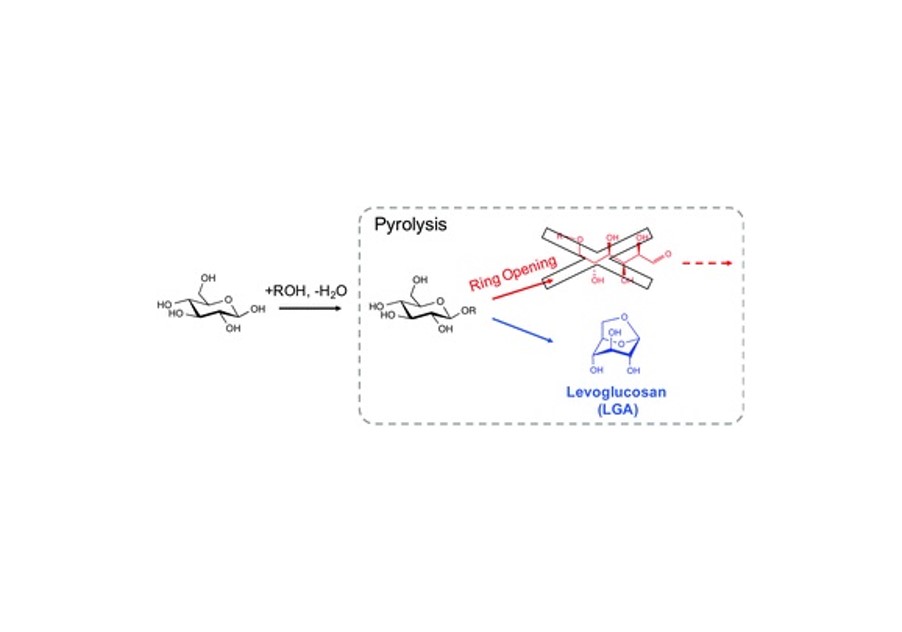
Biomass-derived carbohydrates are an abundant but underutilized carbon source for the production of fuels, chemicals and materials. Pyrolysis is a widely studied thermal process for carbohydrate conversion, but its usefulness in synthesis chemistry is limited by the low structural stability and high reactivity of carbohydrates. Anhydrosugars are primarily prepared from carbohydrate pyrolysis. Their fixed conformation, protected stereoselectivity, and retention of most of their chemical functionality make these sugar derivatives highly desirable precursors. As a model anhydrosugar, LGA is a chiral building block to antibiotics, antiparasitic agents, and other biologically active compounds. It also has strong potential as a sugar-based biorefinery feedstock chemical, with the US Department of Energy identifying it as a top-15 candidate. Large-scale production of LGA and other anhydrosugars remains elusive, however. Our work has been to focus on understanding how to control carbohydrate pyrolysis chemistry (carrying out the dehydration step that leads to intramolecular C1–O6 bond formation; de-selecting the other dehydration pathways, and avoiding ring-opening) through pre- and post-pyrolysis modification of the sugar precursor.
Selected Publications
L. Chen, J. Zhao, S. Pradhan, B. E. Brinson, G. E. Scuseria, Z. C. Zhang, M. S. Wong, "Ring-locking enables selective anhydrosugar synthesis from carbohydrate pyrolysis" Green Chemistry, (2016) DOI: 10.1039/C6GC01600F (Abstract)