2021 Abstracts.
169. M. Long, W.C. Elias, K.N. Heck, Y. Luo, Y.S. Lai, Y. Jin, H. Gu, J. Donoso, T.P. Senftle, C. Zhou, M.S. Wong, and B.E. Rittmann "Hydrodefluorination of Perfluorooctanoic Acid in the H2-Based Membrane Catalyst-Film Reactor with Platinum Group Metal Nanoparticles: Pathways and Optimal Conditions" Enviornmental Science & Technology 55, 24, 16699 - 16707 (2021) DOI: 10.1021/acs.est.1c06528
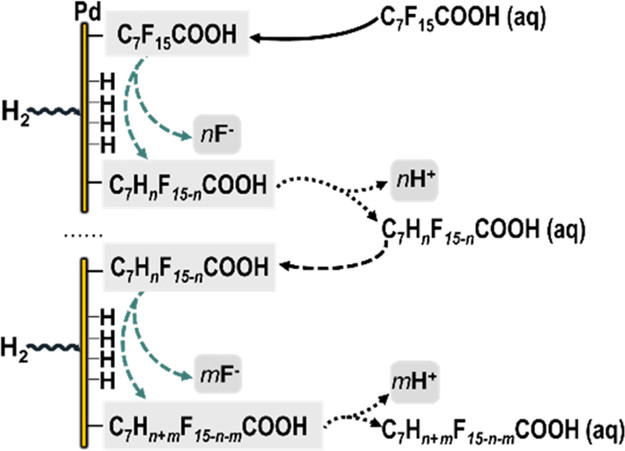
PFAAs (perfluorinated alkyl acids) have become a concern because of their widespread pollution and persistence. A previous study introduced a novel approach for removing and hydrodefluorinating perfluorooctanoic acid (PFOA) using palladium nanoparticles (Pd0NPs) in situ synthesized on H2-gas-transfer membranes. This work focuses on the products, pathways, and optimal catalyst conditions. Kinetic tests tracking PFOA removal, F– release, and hydrodefluorination intermediates documented that PFOA was hydrodefluorinated by a mixture of parallel and stepwise reactions on the Pd0NP surfaces. Slow desorption of defluorination products lowered the catalyst’s activity for hydrodefluorination. Of the platinum group metals studied, Pd was overall superior to Pt, Rh, and Ru for hydrodefluorinating PFOA. pH had a strong influence on performance: PFOA was more strongly adsorbed at higher pH, but lower pH promoted defluorination. A membrane catalyst-film reactor (MCfR), containing an optimum loading of 1.2 g/m2 Pd0 for a total Pd amount of 22 mg, removed 3 mg/L PFOA during continuous flow for 90 days, and the removal flux was as high as 4 mg PFOA/m2/d at a steady state. The EPA health advisory level (70 ng/L) also was achieved over the 90 days with the influent PFOA at an environmentally relevant concentration of 500 ng/L. The results document a sustainable catalytic method for the detoxification of PFOA-contaminated water.
168. C. Lee, S. Guo, H. Rho, J. Levi, S. Garcia-Segura, M.S. Wong, J. Gardea-Torresdey, and P. Westerhoff "Unified Metallic Catalyst Aging Strategy and Implications for Water Treatment" Enviornmental Science & Technology 55, 16, 11284 - 11293 (2021) DOI: 10.1021/acs.est.1c02364
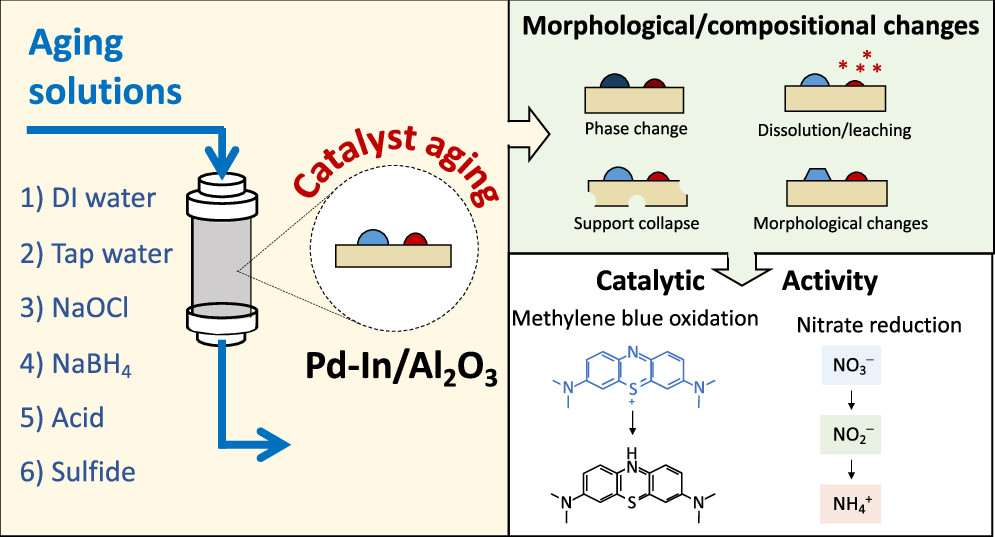
Heterogeneous catalysis holds great promise for oxidizing or reducing a range of pollutants in water. A well-recognized, but understudied, barrier to implement catalytic treatment centers around fouling or aging over time of the catalyst surfaces. To better understand how to study catalyst fouling or aging, we selected a representative bimetallic catalyst (Pd–In supported on Al2O3), which holds promise to reduce nitrate to innocuous nitrogen gas byproducts upon hydrogen addition, and six model solutions (deionized water, sodium hypochlorite, sodium borohydride, acetic acid, sodium sulfide, and tap water). Our novel aging experimental apparatus permitted single passage of each model solution, separately, through a small packed-bed reactor containing replicate bimetallic catalyst “beds” that could be sacrificed weekly for off-line characterization to quantify impacts of fouling or aging. The composition of the model solutions led to the following gradual changes in surface composition, morphology, or catalytic reactivity: (i) formation of passivating species, (ii) decreased catalytic sites due to metal leaching under acid conditions or sulfide poisoning, (iii) dissolution and/or transformation of indium, (iv) formation of new catalytic sites by the introduction of an additional metallic element, and (v) oxidative etching. The model solution water chemistry captured a wide range of conditions likely to be encountered in potable or industrial water treatment. Aging-induced changes altered catalytic activity and provided insights into potential strategies to improve long-term catalyst operations for water treatment.
167. P. Dias da Silva, M.S. Wong, and K. Zygourakis "JP-8 Desulfurization by CuNa-Y Zeolite at Elevated Temperatures Has Two Distinct Stages: Chemisorption Followed by Surface Reactions" Industrial & Engineering Chemistry Research 60, 40, 14534 - 14546 (2021) DOI: 10.1021/acs.iecr.1c02131
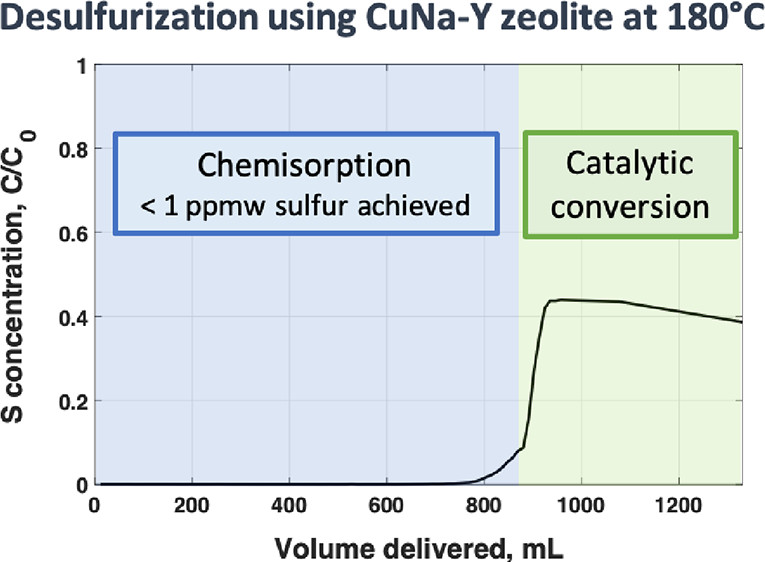
This study evaluates the performance of continuous-flow adsorbers for adsorptive desulfurization. JP-8 fuel with 2230 ppmw of sulfur was treated in a flow-through adsorber packed with CuNa-Y zeolite pellets and operating at 180 °C and 200 psig with liquid hourly space velocities (LHSV) from 0.13 to 3.24 h–1. Our results showed that a flow-through adsorber operating under these conditions can effectively reduce the sulfur content of JP-8 to ultralow values (1–10 ppmw) over the entire LHSV range tested, although the overall performance of the adsorber declined with increasing flow rates as expected. We also observed that the total sulfur removal exceeded the theoretical adsorption limit of our zeolite adsorbent. Detailed characterization of the treated fuel and spent adsorbent via chromatographic and surface analysis techniques revealed that desulfurization occurs in two stages. Sulfur is initially removed via adsorption (chemisorption) on the CuNa-Y zeolite, an assertion supported by simulations with a transient heterogeneous model. As the adsorbent becomes saturated, however, surface chemical reactions start taking place, leading to the formation of hydrogen sulfide and polymerization products and depositing carbon residues on the zeolite. The spent adsorbent was regenerated by treating it with air at 550 or 600 °C, which restored the adsorption capacity of the material to about 90% of its initial value.
166. T.K. Rogers, S. Guo, L. Arrazolo, S. Garcia-Segura, M.S. Wong, and R. Verduzco "Catalytic Capacitive Deionization for Adsorption and Reduction of Aqueous Nitrate" ACS ES&T Water 1, 10, 2233 - 2241 (2021) DOI: 10.1021/acsestwater.1c00195
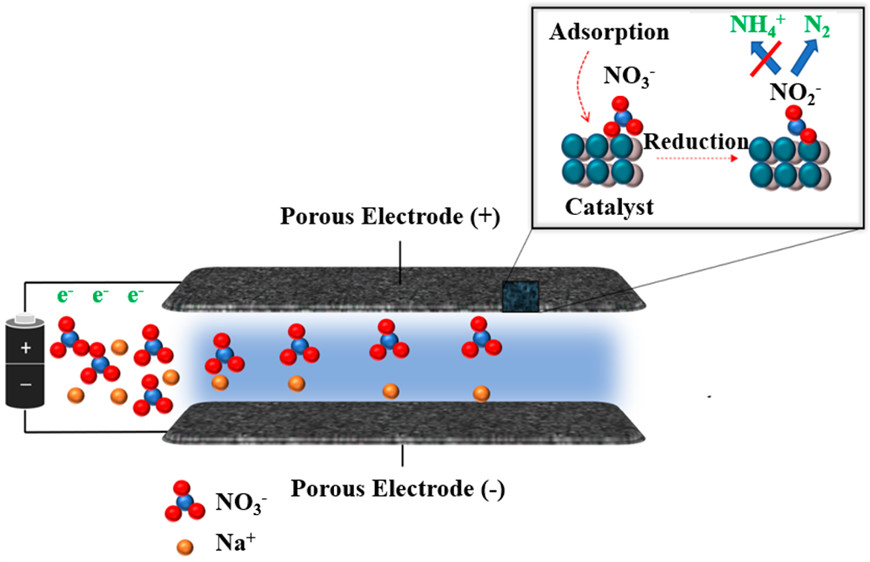
Capacitive deionization (CDI) is an environmental remediation technology that can remove aqueous ionic contaminants, but a major drawback of this process is the production of concentrated waste streams that necessitate further processing. We report a novel approach termed catalytic capacitive deionization (CCDI) that combines selective heterogeneous catalysis with CDI to significantly reduce undesired waste products. In CCDI, porous electrodes containing catalytic nanoparticles are used to electrostatically adsorb and degrade ionic contaminants to less toxic byproducts. We demonstrate CCDI through the removal and reduction of nitrate in water toward innocuous nitrogen gas rather than thermodynamically favorable ammonium. A 50 mg-N/L nitrate was continuously fed to the CCDI reactor at 1 mL/min and adsorbed and treated through 20-min adsorption/catalytic cycles, resulting in an effluent containing 2 mg-N/L nitrate and trace amounts of byproducts, below maximum concentration levels defined for drinking purposes. At a cell potential of 1.5 V, the CCDI system removed 92% of the nitrate in the feed, and 91% of the adsorbed nitrate was selectively transformed to produce innocuous N2. This work demonstrates that CCDI can potentially be used as a novel approach to treat a wide range of aqueous ionic contaminants, significantly advancing the next generation of CDI systems.
165. M. Long, J. Donoso, M. Bhati, W.C. Elias, K.N. Heck, Y. Luo, Y.S. Lai, H. Gu, T.P. Senftle, C. Zhou, M.S. Wong, and B.E. Rittmann "Adsorption and Reductive Defluorination of Perfluorooctanoic Acid over Palladium Nanoparticles" Enviornmental Science & Technology 55, 21, 14836 - 14843 (2021) DOI: 10.1021/acs.est.1c03134
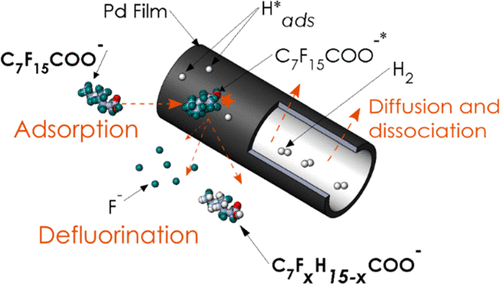
Per- and polyfluoroalkyl substances (PFASs) comprise a group of widespread and recalcitrant contaminants that are attracting increasing concern due to their persistence and adverse health effects. This study evaluated removal of one of the most prevalent PFAS, perfluorooctanoic acid (PFOA), in H2-based membrane catalyst-film reactors (H2-MCfRs) coated with palladium nanoparticles (Pd0NPs). Batch tests documented that Pd0NPs catalyzed hydrodefluorination of PFOA to partially fluorinated and nonfluorinated octanoic acids; the first-order rate constant for PFOA removal was 0.030 h–1, and a maximum defluorination rate was 16 μM/h in our bench-scale MCfR. Continuous-flow tests achieved stable long-term depletion of PFOA to below the EPA health advisory level (70 ng/L) for up to 70 days without catalyst loss or deactivation. Two distinct mechanisms for Pd0-based PFOA removal were identified based on insights from experimental results and density functional theory (DFT) calculations: (1) nonreactive chemisorption of PFOA in a perpendicular orientation on empty metallic surface sites and (2) reactive defluorination promoted by physiosorption of PFOA in a parallel orientation above surface sites populated with activated hydrogen atoms (Hads*). Pd0-based catalytic reduction chemistry and continuous-flow treatment may be broadly applicable to the ambient-temperature destruction of other PFAS compounds.
164. I.S. Nam, X. Verykios, and M.S. Wong "In Memoriam: Maria Flytzani-Stephanopoulos" Applied Catalysis B: Environmental 299, 120653 (2021) DOI: 10.1016/j.apcatb.2021.120653
The Editors, Associate Editors and Editorial Board of APCAT B present this special issue in memory of Maria Flytzani-Stephanopoulos and for celebrating her scientific achievements and impact. We thank her for leading and inspiring us with warmth and grace all these years. We are also thankful to so many who responded positively in submitting their excellent works, even when the world was turned upside down by the COVID-19 pandemic. In all, this special issue contains 46 articles, covering a large spectrum of the environmental catalysis field, particularly single-atom catalysts, metal-support interactions, energy transformations and CO2 utilization, NOx, SOx and VOC. elimination and photocatalytic processes.
163. P. Westerhoff, P.J.J. Alvarez, J. Kim, Q. Li, A. Alabastri, N.J. Halas, D. Villagran, J. Zimmerman, and M.S. Wong "Utilizing the broad electromagnetic spectrum and unique nanoscale properties for chemical-free water treatment" Current Opinion in Chemical Engineering 33, 110709 (2021) DOI: 10.1016/j.coche.2021.100709
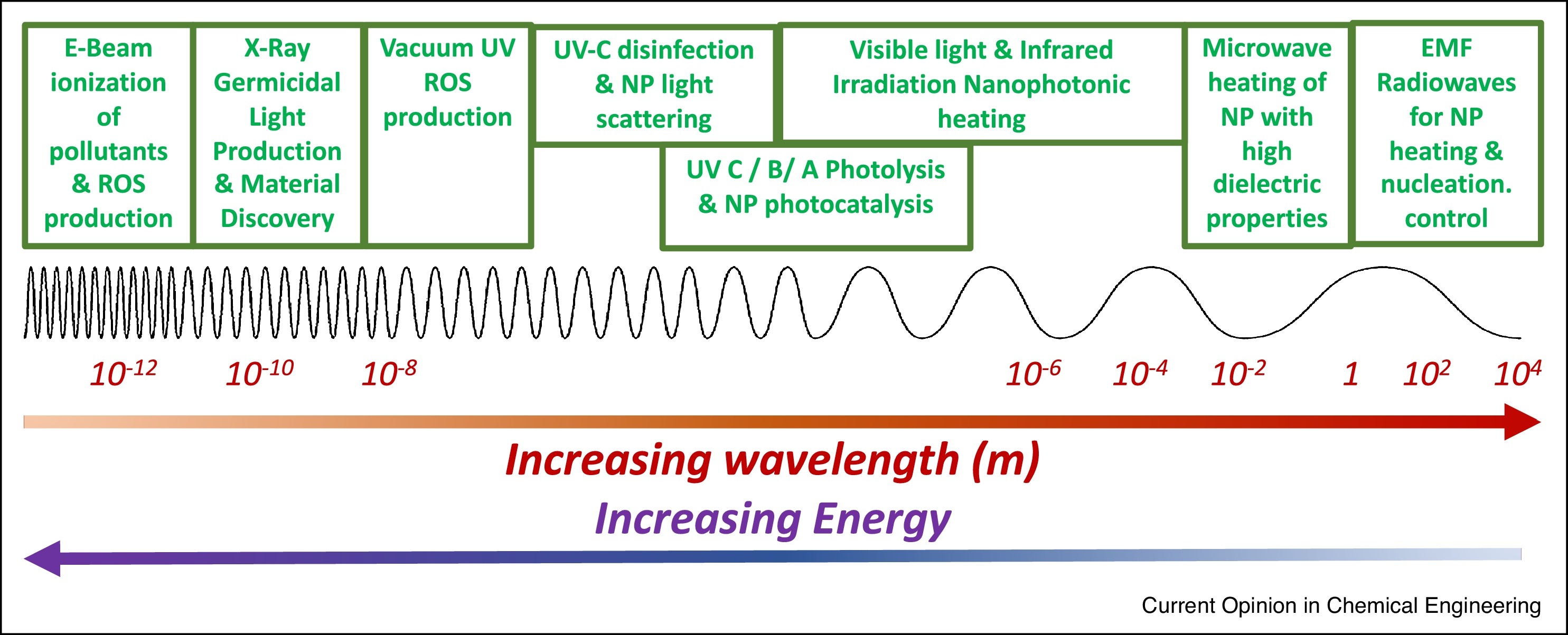
Clean water is critical for drinking, industrial processes, and aquatic organisms. Existing water treatment and infrastructure are chemically intensive and based on nearly century-old technologies that fail to meet modern large and decentralized communities. The next-generation of water processes can transition from outdated technologies by utilizing nanomaterials to harness energy from across the electromagnetic spectrum, enabling electrified and solar-based technologies. The last decade was marked by tremendous improvements in nanomaterial design, synthesis, characterization, and assessment of material properties. Realizing the benefits of these advances requires placing greater attention on embedding nanomaterials onto and into surfaces within reactors and applying external energy sources. This will allow nanomaterial-based processes to replace Victorian-aged, chemical intensive water treatment technologies.
162. M. Kim, A. Peeples, H. Estrada, S. Fowler, L. Chiang, J. Morgan, M. Doganay, B. Walls, B. Wang, J.C. Samba, and M.S. Wong "Destruction of Per- and Polyfluoroalkyl Substances using UVC and Boron Nitride" Waste-Management Education Research Conferences (WERC) (2021) DOI: 10.1109/WERC52047.2021.9477525
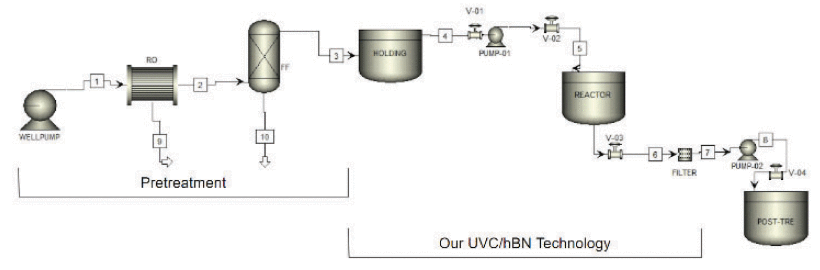
Photocatalytic oxidation is a promising method for degrading per- and polyfluoroalkyl substances (PFAS), but few studies have explored its efficacy in processes beyond the laboratory scale. In this study, degradation of both perfluorooctanoic acid (PFOA) and perfluorooctanesulfonic acid (PFOS) in a bench scale reactor using boron nitride (BN) as a photocatalyst is reported. In addition, a potential full-scale model for incorporation into existing water treatment facilities is proposed. Preliminary results indicated a 53% defluorination rate after 48 h. Final analytical testing indicates 98% PFOA degradation and 79% PFOS degradation after 48 h. Final concentrations of short-chain perfluorocarboxylic acid byproducts are consistent with the previously hypothesized stepwise degradation mechanism for PFOA and suggest that PFOS may degrade through a similar pathway, beginning with the loss of the sulfonic acid head group. There was evidence of NH3 byproduct forming between 48 h and 72 h, suggesting some hydrolytic breakdown of BN occurred during the photocatalysis. BN can carry out destruction of PFOA and PFOS and likely other PFAS compounds, but its stability in water needs to be studied further and improved.
161. S. Sidhik, W. Li, M.H.K. Samani, H. Zhang, Y. Wang, J. Hoffman, A.K. Fehr, M.S. Wong, C. Katan, J. Even, A.B. Marciel, M.G. Kanatzidis, J. Blancon, and A.D. Mohite "Memory Seeds Enable High Structural Phase Purity in 2D Perovskite Films for High-Efficiency Devices" Advanced Materials 33, 29, 2007176 (2021) DOI: 10.1002/adma.202007176
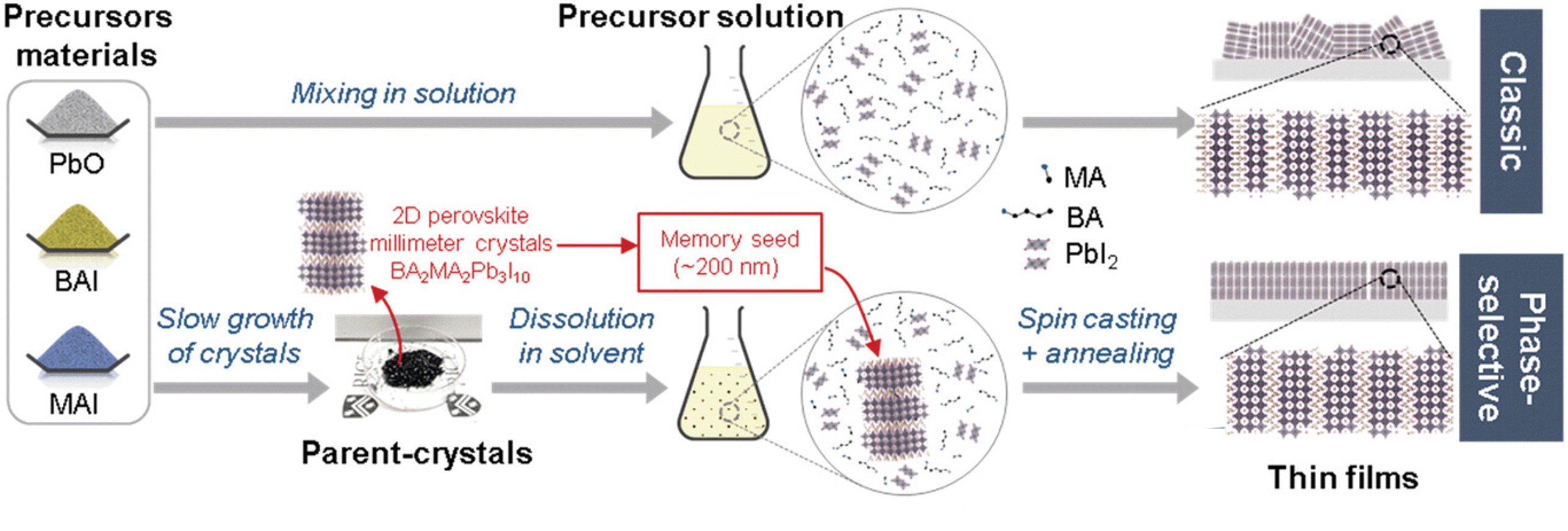
2D perovskites are a class of halide perovskites offering a pathway for realizing efficient and durable optoelectronic devices. However, the broad chemical phase space and lack of understanding of film formation have led to quasi-2D perovskite films with polydispersity in perovskite layer thicknesses, which have hindered device performance and stability. Here, a simple and scalable approach is reported, termed as the “phase-selective method”, to fabricate 2D perovskite thin films with homogenous layer thickness (phase purity). The phase-selective method involves the dissolution of single-crystalline powders with a homogeneous perovskite layer thickness in desired solvents to fabricate thin films. In situ characterizations reveal the presence of sub-micrometer-sized seeds in solution that preserve the memory of the dissolved single crystals and dictate the nucleation and growth of grains with an identical thickness of the perovskite layers in thin films. Photovoltaic devices with a p–i–n architecture are fabricated with such films, which yield an efficiency of 17.1% enabled by an open-circuit voltage of 1.20 V, while preserving 97.5% of their peak performance after 800 h under illumination without any external thermal management.
160. Z. Wu, M. Karamad, X. Yong, Q. Huang, D.A. Cullen, P. Zhu, C. Xia, Q. Xiao, M. Shakouri, F. Chen, J.Y. Kim, Y. Xia, K. Heck, Y. Hu, M.S. Wong, Q. Li, I. Gates, S. Siahrostami, and H. Wang "Electrochemical ammonia synthesis via nitrate reduction on Fe single atom catalyst" Nature Communications 12, 2870 (2021) DOI: 10.1038/s41467-021-23115-x
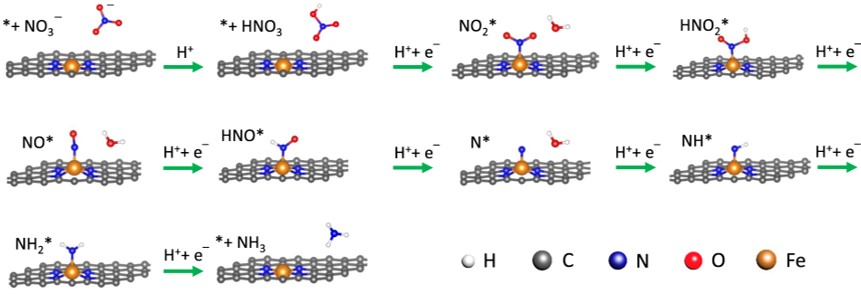
Electrochemically converting nitrate, a widespread water pollutant, back to valuable ammonia is a green and delocalized route for ammonia synthesis, and can be an appealing and supplementary alternative to the Haber-Bosch process. However, as there are other nitrate reduction pathways present, selectively guiding the reaction pathway towards ammonia is currently challenged by the lack of efficient catalysts. Here we report a selective and active nitrate reduction to ammonia on Fe single atom catalyst, with a maximal ammonia Faradaic efficiency of ~ 75% and a yield rate of up to ~ 20,000 μg h−1 mgcat.−1 (0.46 mmol h−1 cm−2). Our Fe single atom catalyst can effectively prevent the N-N coupling step required for N2 due to the lack of neighboring metal sites, promoting ammonia product selectivity. Density functional theory calculations reveal the reaction mechanisms and the potential limiting steps for nitrate reduction on atomically dispersed Fe sites.
159. M. Cokar, J.H. Arredonodo, and M.S. Wong "An Inquiry-Based Learning Undergraduate Laboratory Course During the COVID-19 Pandemic" Chemical Engineering Education 55, 2 (2021)DOI: 10.18260/2-1-370.660-126873

Inquiry based learning (IBL) has gained popularity as a tool for teaching science courses such as chemistry, biology, physics and engineering. This accelerating popularity is driven by technological advancements that allow IBL to be supported in the modern classroom1. The purpose of IBL is to trigger the student’s curiosity and encourage them to ask questions. The student’s
role has become more active with technological advancements and access to multiple sources of information2. In light of this, the chemical and biomolecular engineering department at Rice University has adopted IBL in the undergraduate teaching laboratory courses. We implemented a flipped classroom model where students watched a 15-minute video then took a short quiz on safety and theory before
they attended the first laboratory session. The IBL model focuses on the student to drive the question inquiry process, by engaging and motivating them to evaluate new information pertaining to that knowledge. The laboratory experiment objectives were non-prescriptive allowing the students to develop their own methods for conducting the experiments.
During the spring of 2020, we had all of the IBL course structure in place, and it aided significantly in our shift to “virtual classrooms” after Rice University closed its campus in the wake of the COVID-19 pandemic. By re-designing the undergraduate lab course using IBL principles, we fortuitously gave ourselves built-in flexibility to go to emergency virtual format in Spring 2020, and virtual format in
Fall 2020. This also allows us to go hybrid or full in-person in the future. We provided students with laboratory data so that they could continue the analysis at home. However, students were not able to conduct appropriate experimentation, which is one of the key learning objectives for the laboratory courses.
158. M. Marcos-Hernández, R.A. Arrieta, K. Ventura, J. Hernández, C.D. Powell, A.J. Atkinson, J.S. Markovski, J. Gardea-Torresdey, K.D. Kristovski, P. Westerhoff, M.S. Wong, and D. Villagrán "Superparamagnetic Nanoadsorbents for the Removal of Trace As(III) in Drinking Water" Environmental Advances 4, 100046 (2021) DOI: 10.1016/j.envadv.2021.100046
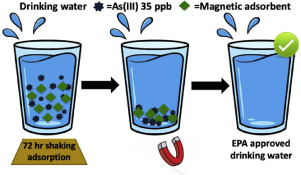
A series of novel zeolitic imidazolate framework (ZIF) decorated superparamagnetic graphene oxide hybrid nanoadsorbents were synthesized, characterized, and tested for their As(III) adsorbed amount in simulated drinking water. The three composite nanomaterials are based each on three isostructural and water stable ZIFs, (C-1 based on ZIF-8, C-2 based on ZIF-67, and C-3 based on ZIF-Zn/Co). The composite nanomaterials and there parent materials were characterized through pXRD, TEM, FTIR, BET and magnetometry methods (SQUID), and were tested as adsorbents in a representative drinking water matrix containing arsenite (As(III)) at an initial trace concentration (realistic in some natural drinking water sources) of 35 µg/L. The nanoadsorbents were magnetically captured and removed after adsorption in batch conditions. Out of the three composites, C-2 shows the highest As(III) adsorbed amount at an initial concentration of 35 µg/L (q0) of 202 µg/g, followed by C-3 with 102 µg/g and C-1 with 82 µg/g.
157. Y. Xu, K.N. Heck, C. Ayala-Orozco, J.H. Arredondo, W. Zenor, M. Shammai, and M.S. Wong "Heavy oil viscosity reduction at mild temperatures using palladium acetylacetonate" Fuel 294, 120546 (2021) DOI: 10.1016/j.fuel.2021.120546
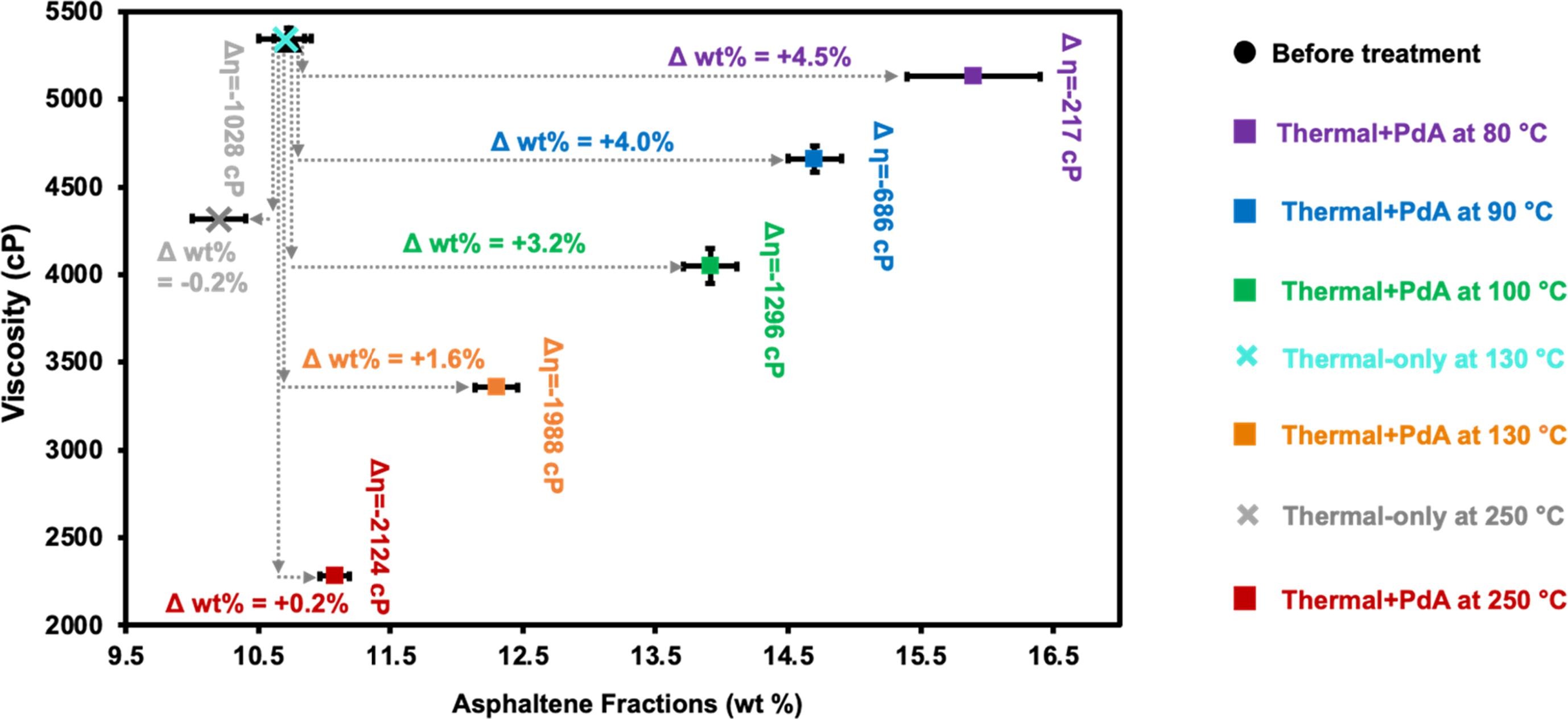
Metal-ligand compounds (“MLCs”) have been shown to reduce heavy oil viscosity and upgrade oil quality. However, MLCs generally require high treatment temperatures (around 250 °C), which is undesirably energy-intensive. We identified palladium(II) acetylacetonate (“PdA”) as a model MLC that can operate at mild temperatures (less than 200 °C). We studied its effectiveness on heavy oil viscosity reduction in the range of 80–300 °C using viscometry, SARA analysis, GC–MS, XPS, and XRD to characterize Peace River oil samples thermally treated with and without PdA. This MLC effectively lowered oil viscosity at all treatment temperatures, whereas thermal-only treatments did not reduce viscosity below 160 °C. The thermal treatment with PdA in the 130–250 °C range reduced viscosity by up to ~35% more than the thermal treatment alone. GC–MS and TGA results indicated the PdA partially decomposed at 80 °C and higher temperatures, releasing acetylacetone (“HA”), which lowered oil viscosity. The temperature and HA effects did not completely account for the observed viscosity reduction from thermal treatment with PdA, indicating there were other significant effects. In the 80–130 °C range, the asphaltene fraction increased due to PdA or its decomposition products intercalating into the asphaltene clusters. At temperatures around 250 °C, the resin fraction decreased, correlating to in situ formed metallic Pd that catalytically hydrogenate the resin sulfonyl groups to aliphatic sulfur. This new understanding of the temperature-dependent impact – acetylacetonate ligand, MLC-asphaltene attraction, and palladium metal catalyst formation – on oil viscosity changes provides an improved approach to developing new MLCs for field-relevant conditions.
156. C.D. Powell, A.W. Lounsbury, Z.S. Fishman, C.L. Conrad, M.J. Gallagher, D. Villagrán, J.B. Zimmerman, L.D. Pfefferle, and M.S. Wong "Nano-structural effects on Hematite (α-Fe2O3) nanoparticle radiofrequency heating" Nano Convergence 8 (2021) DOI: 10.1186/s40580-021-00258-7
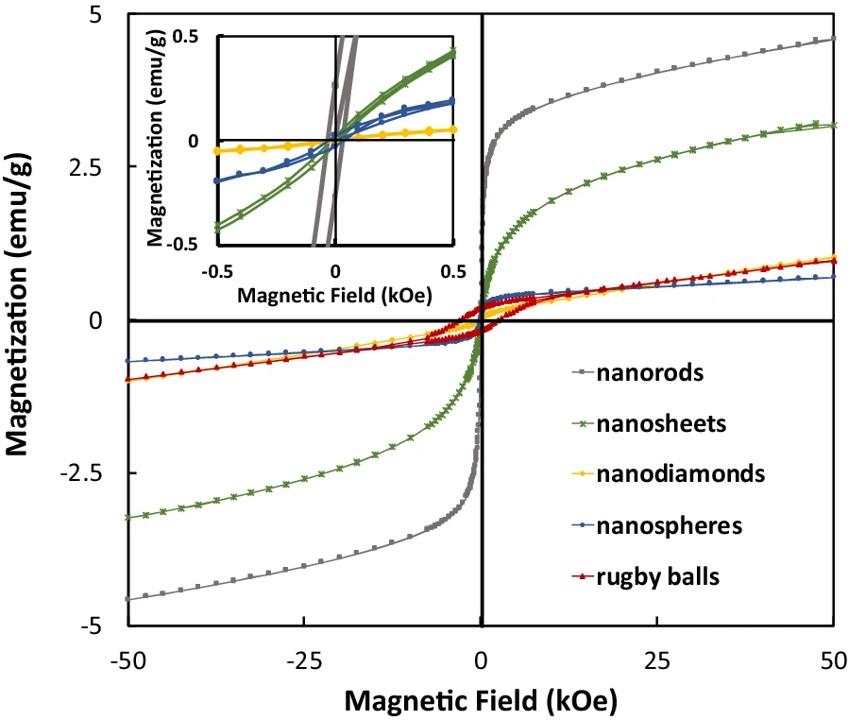
Nano-sized hematite (α-Fe2O3) is not well suited for magnetic heating via an alternating magnetic field (AMF) because it is not superparamagnetic—at its best, it is weakly ferromagnetic. However, manipulating the magnetic properties of nano-sized hematite (i.e., magnetic saturation (Ms), magnetic remanence (Mr), and coercivity (Hc)) can make them useful for nanomedicine (i.e., magnetic hyperthermia) and nanoelectronics (i.e., data storage). Herein we study the effects of size, shape, and crystallinity on hematite nanoparticles to experimentally determine the most crucial variable leading to enhancing the radio frequency (RF) heating properties. We present the synthesis, characterization, and magnetic behavior to determine the structure–property relationship between hematite nano-magnetism and RF heating. Increasing particle shape anisotropy had the largest effect on the specific adsorption rate (SAR) producing SAR values more than 6× greater than the nanospheres (i.e., 45.6 ± 3 W/g of α-Fe2O3 nanorods vs. 6.89 W/g of α-Fe2O3 nanospheres), indicating α-Fe2O3 nanorods can be useful for magnetic hyperthermia.