2020 Abstracts.
155. C.D. Powell, S. Guo, L.M. Godret-Miertschin, K. Ventura, A.W. Lounsbury, C.A. Clark, D. Villagrán, J.B. Zimmerman, A.J. Atkinson, P. Westerhoff, and M.S. Wong "Magnetically recoverable carbon-coated iron carbide with arsenic adsorptive removal properties" SN Applied Sciences 2, 1690 (2020) DOI: 10.1007/s42452-020-03491-7
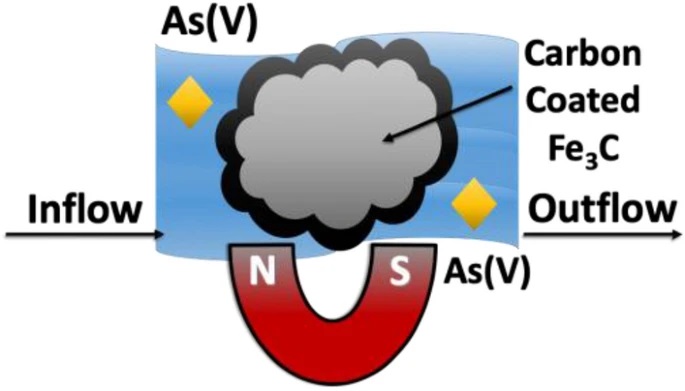
Magnetic particles, generally nanostructured and magnetite-based, have been studied extensively to remove drinking water contaminants. Compositions beyond Fe3O4 could address long-standing issues of magnetic recoverability and materials integrity in drinking waters. Herein carbon-coated iron carbide (Fe3C@C) were studied for the first time for their stability, magnetic characteristics, magnetic separability, and arsenic adsorptive properties. Experimental results show that (i) Fe3C@C with a 9-nm thick graphitic shell is chemically stable in simulated drinking water; (ii) is ferromagnetic with small magnetic remanence and a magnetic saturation that is ~2× greater than Fe3O4; (iii) can be separated from water magnetically under continuous-flow conditions with greater than 99% recovery; and (iv) has a surface area-normalized adsorption capacity for arsenic (6.75 µg/m2) of the same order of magnitude as that of Fe3O4 (9.62 µg/m2). Fe3C@C can be a viable alternative to Fe3O4 with further development, for the magnetic removal of arsenic and other contaminants from drinking water sources.
154. S. Guo, C.D. Powell, D. Villagrán, and M.S. Wong "Magnetic In–Pd catalysts for nitrate degradation" Environmental Science: Nano 7, 2681-2690 (2020) DOI: 10.1039/D0EN00069H
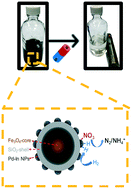
Magnetic catalysts offer the possibility of rapidly eliminating nitrate oxyanions, a ubiquitous drinking water contaminant, without generating a secondary waste stream. Herein, we report the synthesis of a magnetically recoverable bimetallic Pd–In material that exhibits excellent chemical stability, reusability, and high nitrate removal efficiency. This four-component catalyst (Pd–In/nFe3O4@SiO2) contains nanocrystalline magnetite with a silica shell upon which indium-decorated palladium nanoparticles attach. The SiO2 shell slowed down iron leaching from Fe3O4 and the bimetallic nano-domains showed nitrate reduction activity in deionized water without obvious deactivation through multiple recovery and reuse cycles. This magnetically responsive, reusable catalyst, which retained activity in simulated drinking water, can serve as a design basis for materials to degrade other oxyanion water contaminants.
153. T.K. Rogers, P.D. Dongare, A. Alabastri, N.J. Halas, J. Metz, J. Mathieu, P.J.J. Alvarez, L. Carter, M.S. Wong, and R. Verduzco "Nanotechnology for Beyond Earth Water Treatment" International Conference on Environmental Systems (2020) Conference Collection
NASA has embarked on a journey to enable human exploration on the Moon and Mars by 2024. These long duration missions beyond low earth orbit (LEO) will require advanced water treatment and reuse technologies for life support systems to support crew and system needs. Resupply to deep space destinations is not desirable and sustained human presence in a lunar environment increases the necessity for robust and reliable systems. To reduce propulsion costs and transit space allocations, mass, power consumption, and volume must be minimized for all systems. Additionally, a beyond (LEO) water treatment system process must be able to tolerate both operational and dormant periods. Herein, we present nanotechnologies developed by the Nanotechnology-Enabled Water Treatment (NEWT) center as advanced solutions to meet the aforementioned requirements. This survey of fit-for-purpose modular technologies includes room temperature nanocatalysis, nanophotonics, nano-selective scalant control, quorum sensing and biofouling control techniques, and nano enabled fluid management. A case study of integrating nanotechnology into state-of-the-art and developing systems is also presented.
152. H. Javed, J. Metz, T.C. Eraslan, J. Mathieu, B. Wang, G. Wu, A. Tsai, M.S. Wong, and P.J.J. Alvarez "Discerning the Relevance of Superoxide in PFOA Degradation" Environmental Science & Technology Letters 7, 9, 653 - 658 (2020) DOI: 10.1021/acs.estlett.0c0505
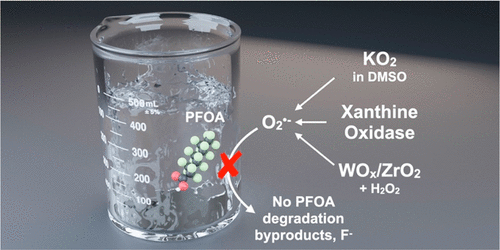
Perfluorooctanoic acid (PFOA) is a widely distributed recalcitrant contaminant. In recent years, advanced oxidation processes have been explored for PFOA degradation, yet factors influencing their efficacy and degradation mechanism are not fully understood. Here, we resolve ambiguity in the literature regarding the role of superoxide in PFOA degradation (e.g., by nucleophilic attack) by considering three pure superoxide-producing systems: KO2 in dimethyl sulfoxide, xanthine oxidase with hypoxanthine, and WOx/ZrO2 catalyst with H2O2. Superoxide production was confirmed in all systems by electron paramagnetic resonance spectroscopy and by precipitation of nitroblue tetrazolium, a common superoxide probe. Positive control experiments showed that the produced superoxide degrades ~48% of bisphenol A within 1 day, corroborating the fact that superoxide was sufficiently stable and available for reaction in the test systems. However, no PFOA degradation was observed, which was corroborated by the absence of fluoride and degradation byproducts in all three systems. Therefore, other reaction pathways should be explored for PFOA degradation.
151. S. Garcia-Segura, X. Qu, P.J.J. Alvarez, B.P. Chaplin, W. Chen, J.C. Crittenden, Y. Feng, G. Gao, Z. He, C. Hou, Z. Hu, G. Jiang, J. Kim, J. Li, Q. Li, J. Ma, J. Ma, A.B. Nienhauser, J. Niu, B. Pan, X. Quan, F. Rozani, D. Villagran, T.D. Waite, W.S. Walker, C. Wang, M.S. Wong, and P. Westerhoff "Opportunities for nanotechnology to enhance electrochemical treatment of pollutants in potable water and industrial wastewater – a perspective" Environmental Science: Nano 7, 2178-2194 (2020) DOI: 10.1039/D0EN00194E
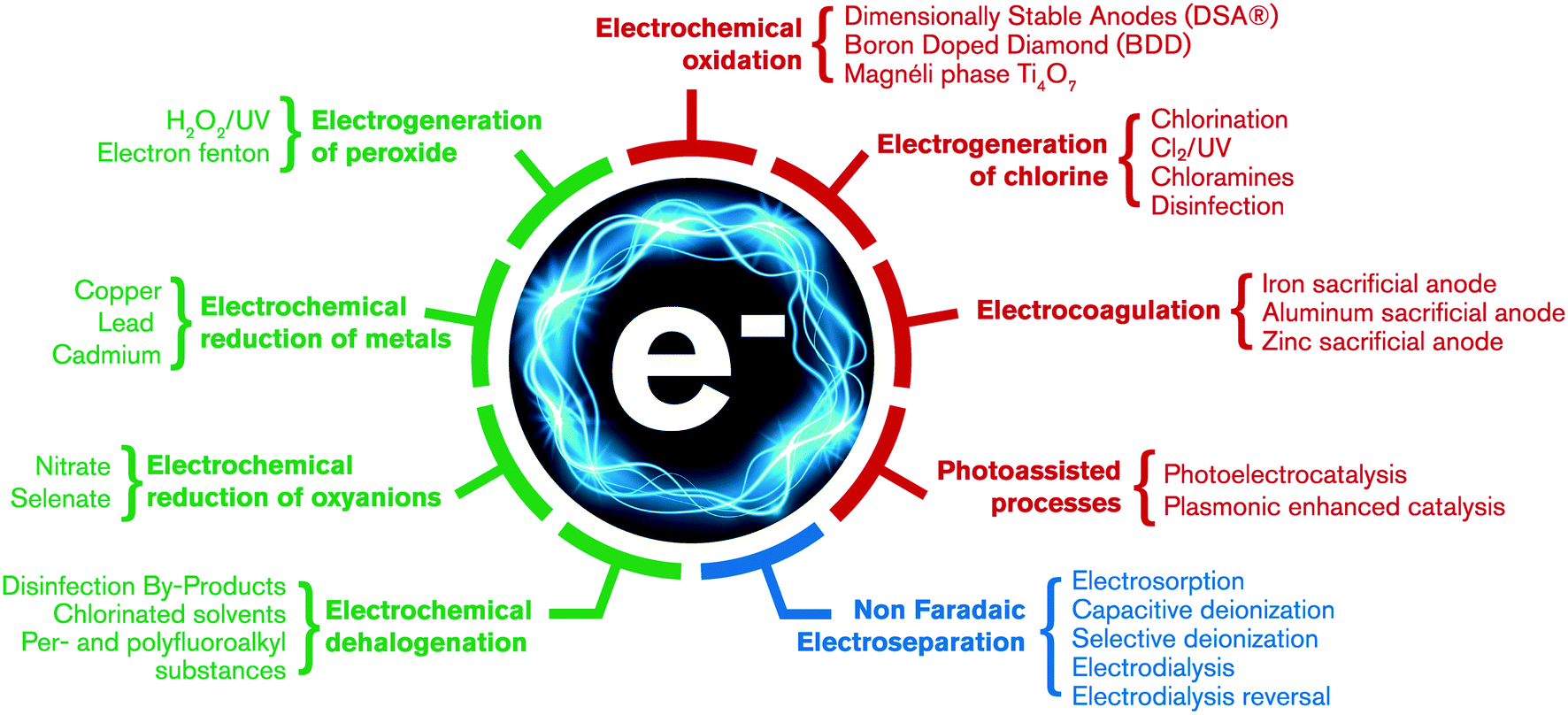
Based upon an international workshop, this perspective evaluates how nano-scale pore structures and unique properties that emerge at nano- and sub-nano-size domains could improve the energy efficiency and selectivity of electroseparation or electrocatalytic processes for treating potable or waste waters. An Eisenhower matrix prioritizes the urgency or impact of addressing potential barriers or opportunities. There has been little optimization of electrochemical reactors to increase mass transport rates of pollutants to, from, and within electrode surfaces, which become important as nano-porous structures are engineered into electrodes. A “trap-and-zap” strategy is discussed wherein nanostructures (pores, sieves, and crystal facets) are employed to allow localized concentration of target pollutants relative to background solutes (i.e., localized pollutant trapping). The trapping is followed by localized production of tailored reactive oxygen species to selectively degrade the target pollutant (i.e., localized zapping). Frequently overlooked in much of the electrode-material development literature, nano-scale structures touted to be highly “reactive” towards target pollutants may also be the most susceptible to material degradation (i.e., aging) or fouling by mineral scales that form due to localized pH changes. A need exists to study localized pH and electric-field related aging or fouling mechanisms and strategies to limit or reverse adverse outcomes from aging or fouling. This perspective provides examples of the trends and identifies promising directions to advance nano-materials and engineering principles to exploit the growing need for near chemical-free, advanced oxidation/reduction or separation processes enabled through electrochemistry.
150. L. Duan, B. Wang, K. Heck, S. Guo, C.A. Clark, J. Arredondo, M. Wang, T.P. Senftle, P. Westerhoff, X. Wen, Y. Song, and M.S. Wong "Efficient Photocatalytic PFOA Degradation over Boron Nitride" Environmental Science & Technology Letters 7, 8, 613-619 (2020) DOI: 10.1021/acs.estlett.0c00434
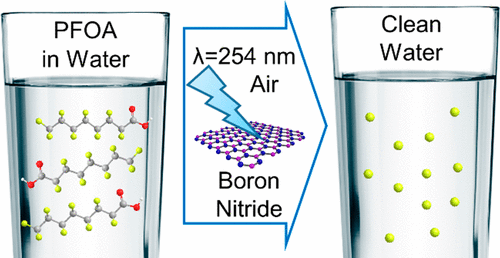
Concern over water contamination by per/polyfluoroalkyl substances (PFAS) has highlighted the lack of effective treatment approaches. Photocatalysis offers advantages of using ambient conditions for reaction, air as the oxidant, and light as the energy source, but identifying photoactive materials is challenging. Herein, we report that boron nitride (BN) degrades PFOA upon irradiation with 254 nm light. The ability of BN to degrade PFOA photocatalytically has previously been unreported and is unexpected, because its band gap is too large for light absorption. On the basis of scavenger results, we suggest that PFOA degrades in the presence of BN via a hole-initiated reaction pathway similar to the TiO2 case and involves superoxide/hydroperoxyl and hydroxyl radicals. We surmised that defects allow BN to absorb in the UVC range and to photogenerate reactive oxygen species. Sealed batch studies indicated BN was ~2 and ~4 times more active than TiO2, before and after ball milling the material, respectively. BN can be reused, showing no decrease in activity over three cycles. BN was active for the photocatalytic degradation of GenX, another PFAS of concern. These findings present fresh opportunities for materials design and for the re-evaluation of other wide band gap semiconductors for PFAS photocatalytic degradation.
149. K. Ventura, R.A. Arrieta, M. Marcos-Hernández, V. Jabbari, C.D Powell, R. Turley, A.W. Loundsbury, J.B. Zimmerman, J. Gardea-Torresdey, M.S. Wong, and D. Villagrán "Superparamagnetic MOF@GO Ni and Co based hybrid nanocomposites as efficient water pollutant adsorbents" Science of the Total Environment 738, 139213 (2020) DOI: 10.1016/j.scitotenv.2020.139213
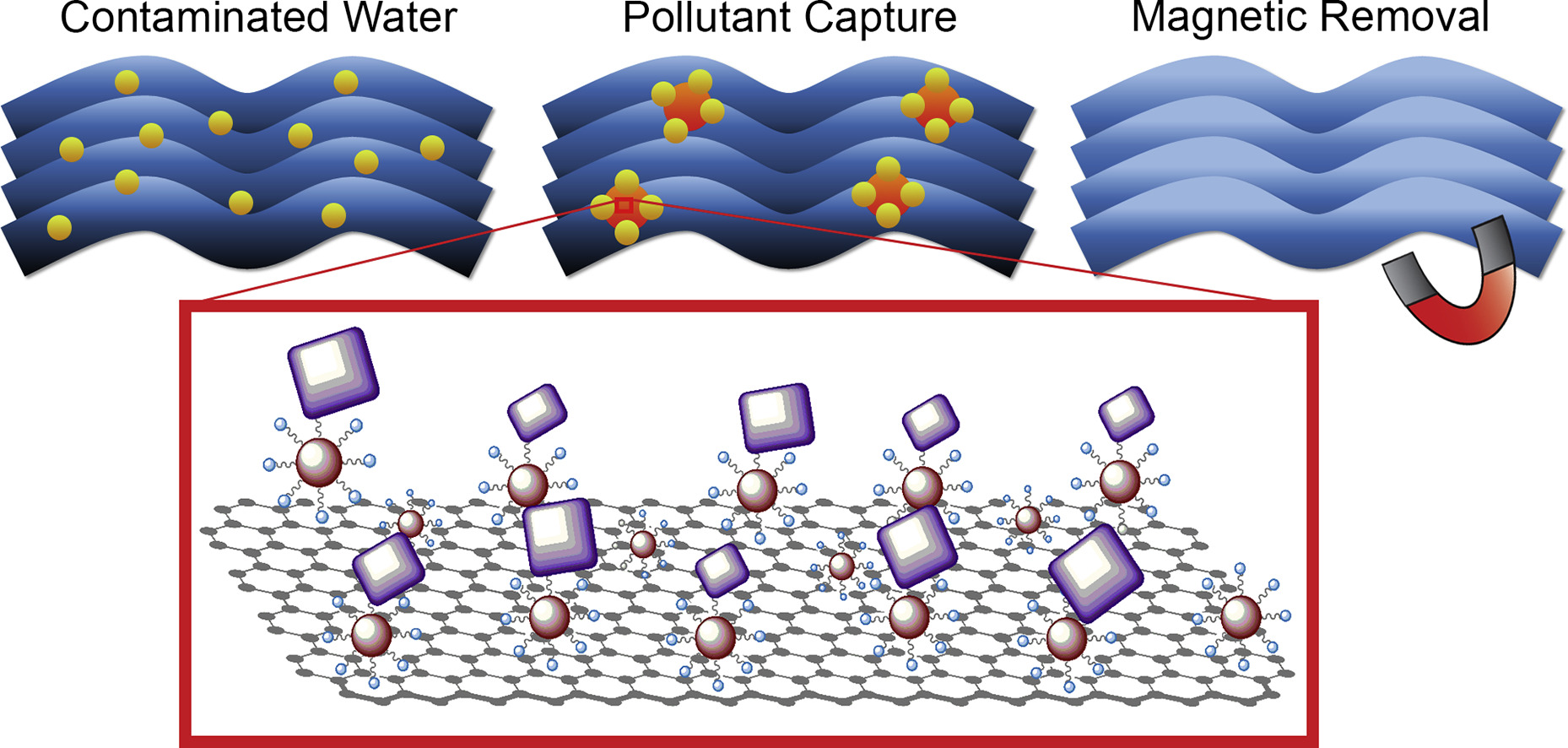
A series of highly efficient adsorbents were developed using Ni3(BTC)2 and Co3(BTC)2 metal-organic frameworks (MOFs) and Fe3O4 magnetic nanoparticles (MNPs) to functionalize graphene oxide (GO). XRD results show high crystallinity of the prepared nanomaterials and the successful decoration of Ni3(BTC)2 and Co3(BTC)2 MOFs over the GO substrate (BTC = benzene-1,3,5-tricarboxylic acid). SEM and TEM imaging show the successful formation of nanoscale MOFs and Fe3O4 MNPs over GO. IR spectroscopy supports the characterization and successful preparation of the Fe3O4/MOF@GO hybrid composite nanoadsorbents. The prepared composite nanoadsorbents were used to sorb Methylene Blue (MB) as a model for common organic pollutants in water and common ions (Na+, Ca2+, Mg2+, SO42−, SiO32−) from a brackish water model. The adsorbed concentration at equilibrium of MB of the prepared composite nanoadsorbents increases by an average of 30.52 and 13.75 mg/g for the Co and Ni composite, respectively, when compared to the MOFs parent materials. The adsorbed amount of sulfate ions increases by 92.1 mg/g for the Co composite and 112.1 mg/g for the Ni composite, when compared to graphene oxide. This adsorption enhancement is attributed to suppressed aggregation through increased dispersive forces in the MOFs due to the presence of GO, formation of nanoscale MOFs over the GO platform, and the hindering of stacking of the graphene layers by the MOFs. Leaching tests show that the release of Co and Ni ions to water is reduced from 105.2 and 220 mg/L, respectively, in the parent MOF materials to 0.5 and 16.4 mg/L, respectively, in the composite nanoadsorbents. These findings show that the newly developed composite nanoadsorbents can sorb organic pollutants, and target sulfate and silicate anions, which makes them suitable candidates for water and wastewater treatments.
148. W.C. Elias, K.N. Heck, S. Guo, S. Yazdi, C. Ayala-Orozco, S. Grossweiler, J.B. Domingos, E. Ringe, and M.S. Wong "Indium-decorated Pd nanocubes degrade nitrate anions rapidly" Applied Catalysis B: Environmental 276, 119048 (2020) DOI: 10.1016/j.apcatb.2020.119048
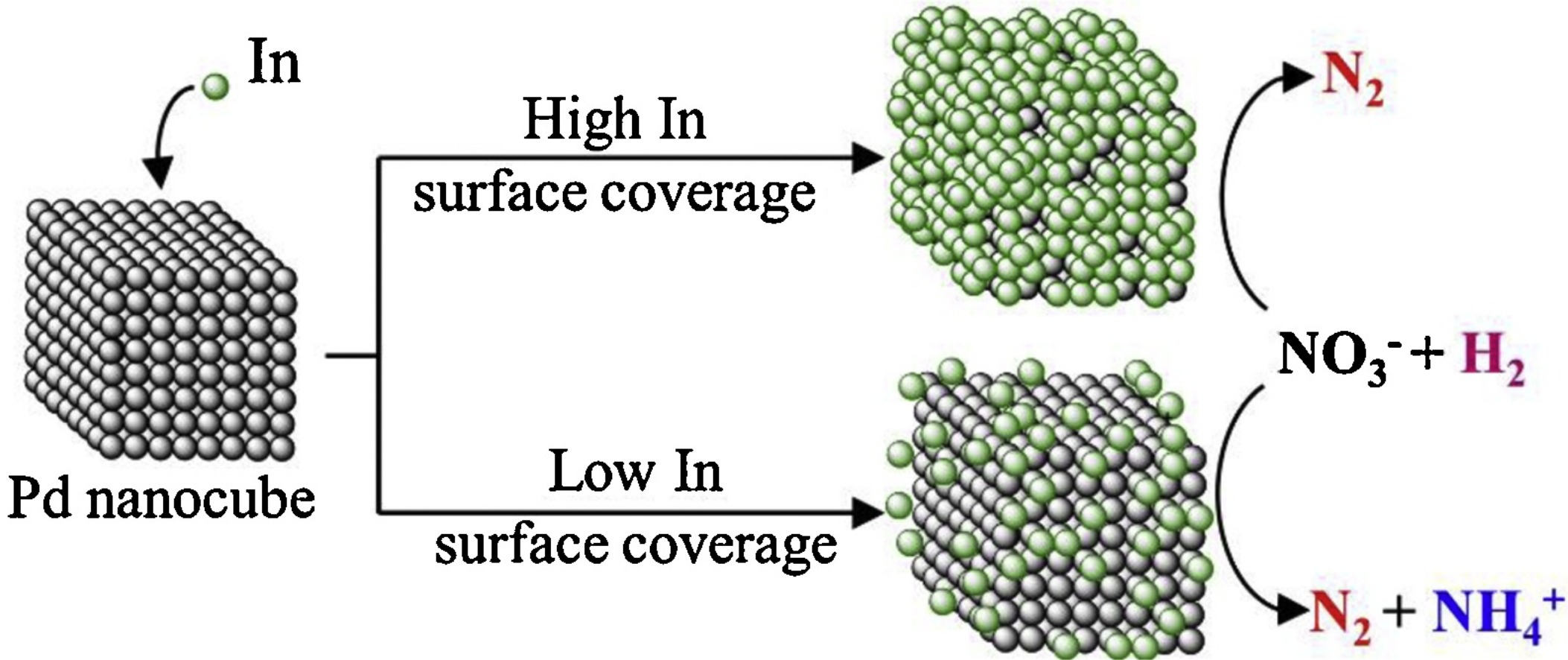
Indium-decorated palladium nanoparticles (In-on-PdNPs) are active for room-temperature catalytic reduction of aqueous nitrate, where the active sites are metallic In atoms on the Pd surface. The PdNPs are pseudo-spherical in shape, and it is unclear if their faceted nature plays a role in nitrate reduction. We synthesized different-sized, cube-shaped NPs with differing In coverages (sc%), and studied the resultant In-on-Pd-nanocubes (NCs) for nitrate reduction. The NCs exhibited volcano-shape activity dependence on In sc%, with peak activity around 65–75 sc%. When rate constants were normalized to undercoordinated atoms (at edge + corners), the NCs exhibited near-identical maximum activity (20×-higher than In-on-PdNPs) at ρIn/Pd edge+corner ~0.5 (~5 In atoms per 10 edge and corner atoms). NCs with a higher In edge + corner density (ρIn/Pd edge+corner ~1.5) were less active but did not generate NH4+ at nitrate conversions tested up to 36%. Edge-decorated cubes may be the structural basis of improved bimetallic catalytic denitrification of water.
147. Y.B. Yin, C.L. Conrad, K.N. Heck, I.A. Said, C.D. Powell, S. Guo, M.A. Reynolds, and M.S. Wong "Room-Temperature Catalytic Treatment of High-Salinity Produced Water at Neutral pH" Industrial & Engineering Chemistry Research 59, 22, 10356-10363 (2020) DOI: 10.1021/acs.iecr.0c00521
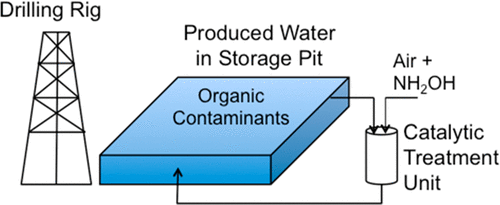
Produced waters from hydraulic fracturing (HFPW) operations greatly challenge traditional water treatment technologies due to the high concentrations of total dissolved solids (TDS), highly complex and variable water matrices, and significant residual hydrocarbon content. We recently reported the unusual ability of a PdAu catalyst to degrade phenol in simulated HFPW at room temperature by generating H2O2 in situ from formic acid and air. Phenol removal occurred at TDS levels as high as ~10 000 ppm (ionic strength I = 0.3 M), but the catalytic reaction required pH less than 4 to proceed. Here, we find that PdAu, Pd, and Au degraded phenol in the pH 5–8 range by using hydroxylamine as the hydrogen source in place of formic acid. Pd exhibited the highest activity, and Au the least. Activity of the monometallic catalysts decreased >70% as TDS increased from 0 to ~100 000 ppm (I = 3 M), whereas the PdAu was comparatively less affected (~50% activity decrease). All catalysts remained active at TDS levels as high as 100 000 ppm. The majority of the hydroxylamine formed N2, however this reaction generated additional nitrite/nitrate anion byproducts with nitrogen selectivities ranging from 0.5% to 11.5%, depending on the catalyst identity and reaction salinity. To demonstrate one possible flow treatment process concept, we constructed and tested a recirculating trickle bed reactor that removed 28% phenol from simulated HFPW over 48 h. These results show the potential of oxidation catalysis as a treatment approach for produced water and other high-salinity industrial wastewaters.
146. X. Wang, M. Sun, Y. Zhao, C. Wang, W. Ma, M.S. Wong, and M. Elimelech "In Situ Electrochemical Generation of Reactive Chlorine Species for Efficient Ultrafiltration Membrane Self-Cleaning" Environmental Science & Technology 54, 11, 6997-7007 (2020) DOI: 10.1021/acs.est.0c01590
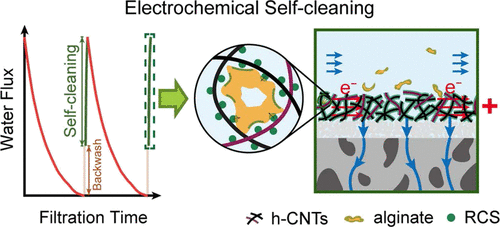
Reactive membranes based on hydroxyl radical generation are hindered by the need for chemical dosing and complicated module and material design. Herein, we utilize an electrochemical approach featuring in situ generation of reactive (radical) chlorine species (RCS) through anodization of chloride ions for membrane self-cleaning. A hybridized carbon nanotube (CNT) functionalized ceramic membrane (h-CNT/CM), possessing high hydrophilicity, permeability, and conductivity, was fabricated. Using carbamazepine (CBZ) as a probe, we confirmed the presence of RCS in the electrified h-CNT/CM. The rapid and complete degradation of CBZ in a single-pass ultrafiltration indicates high localized RCS concentration within the three-dimensional porous CNT interwoven layer. We further demonstrate that the electro-generation of RCS is a critical pre-step for free chlorine (HClO and ClO−) formation. Self-cleaning efficiency of the membrane after fouling with a model organic foulant (alginate) was assessed using an electrified cross-flow membrane filtration system. The fouled h-CNT/CM exhibits a near complete water flux recovery following a short (one minute) self-cleaning with an applied voltage of 3 or 4 V and feed solutions of 100 or 10 mM sodium chloride, respectively. Considering the superior performance of the RCS-mediated self-cleaning compared to conventional membrane chemical cleaning using sodium hypochlorite, our results exemplify an effective strategy for in situ electro-generation of RCS to achieve highly-efficient membrane self-cleaning.
145. Y. Xu, C. Ayala-Orozco, P. Chiang, M. Shammai, and M.S. Wong "Understanding the role of iron (III) tosylate on heavy oil viscosity reduction" Fuel 274, 117808 (2020) DOI: 10.1016/j.fuel.2020.117808
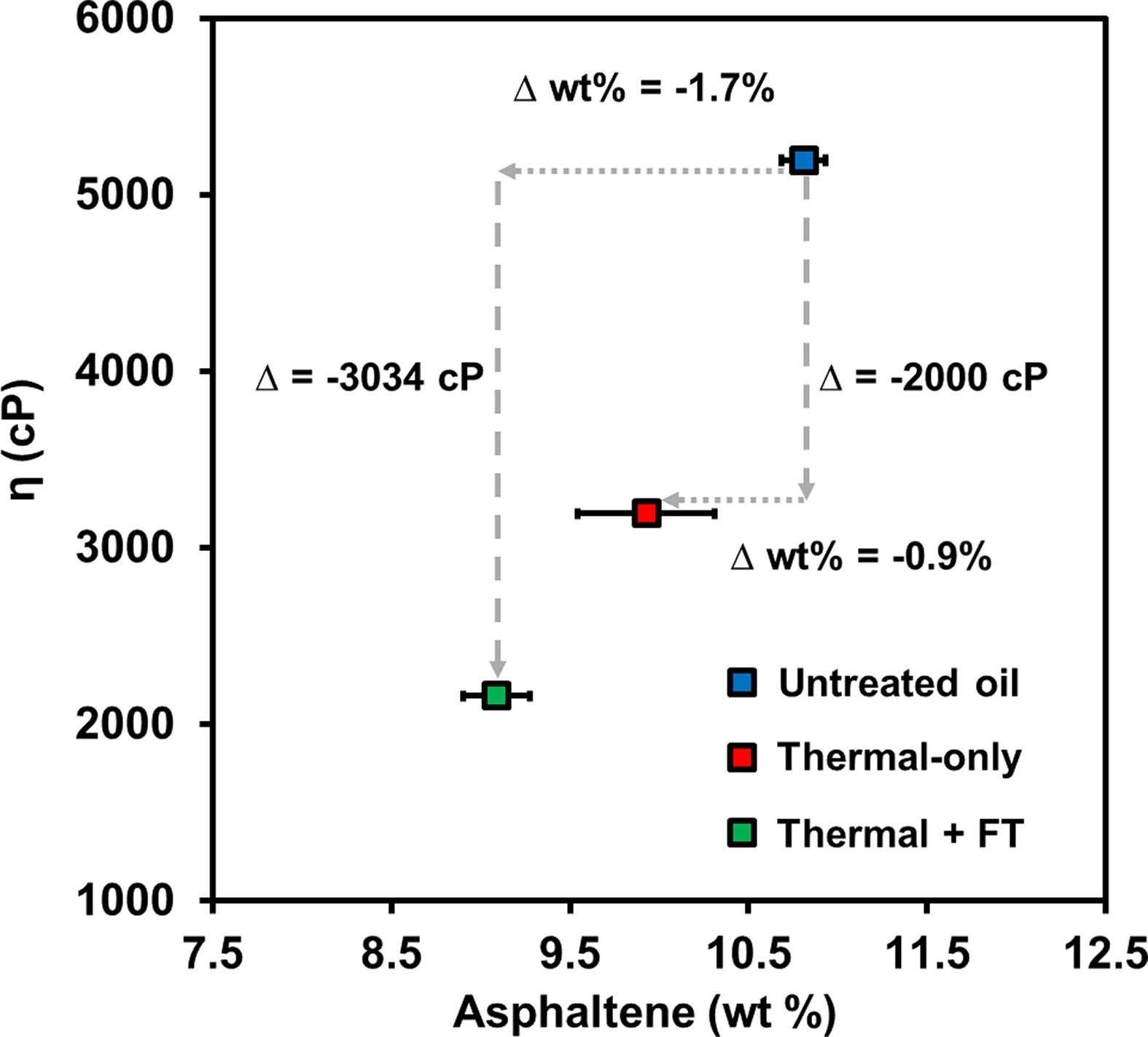
Heavy oil is an abundant energy resource, but its recovery remains challenging primarily due to its high viscosity. Thermally enhanced oil recovery in the presence of metal-ligand compounds (MLCs) has been studied as a promising method for in situ viscosity reduction and oil quality upgrading. In spite of its importance, the interactions between MLCs and crude oil components at the molecular-level are poorly understood, and their mechanistic details for viscosity reduction are unclear. Here we studied viscosity changes of Peace River oil in the range of 80–295°C in the presence and absence of iron (III) tosylate (para-toluenesulfonate) MLC and analyzed reaction products via viscometry, TGA-MS, XPS, GC-MS, SARA analysis, and elemental analysis. Whereas thermal treatment lowered viscosity at 190°C and above, thermal treatment with the iron tosylate MLC decreased viscosity only at temperatures above 230°C. The MLC effect was most substantial at 280°C, at which viscosity decreased by 58% (compared to 39% in absence of the MLC at the same temperature). The MLC likely lowered oil viscosity by catalytically reacting with the asphaltene to decrease its total content in oil, and by releasing a ligand to form 4-methylbenzenethiol (MBT) that interfered with asphaltene intermolecular interactions. At temperatures below 230°C, the MLC unexpectedly raised oil viscosity, likely due to bridging interactions with asphaltene. This understanding of MLC-induced deviscosification provides a selection rationale for appropriate metals and ligands for enhancing heavy oil recovery.
144. R. Verduzco and M.S. Wong "Fighting PFAS with PFAS: Charged and Partially Fluorinated Hydrogels are Demonstrated to be a Promising New Approach to Selectively Uptake PFAS Contaminants in Water" ACS Central Science 6, 4, 453-455 (2020) DOI: 10.1021/acscentsci.0c00164
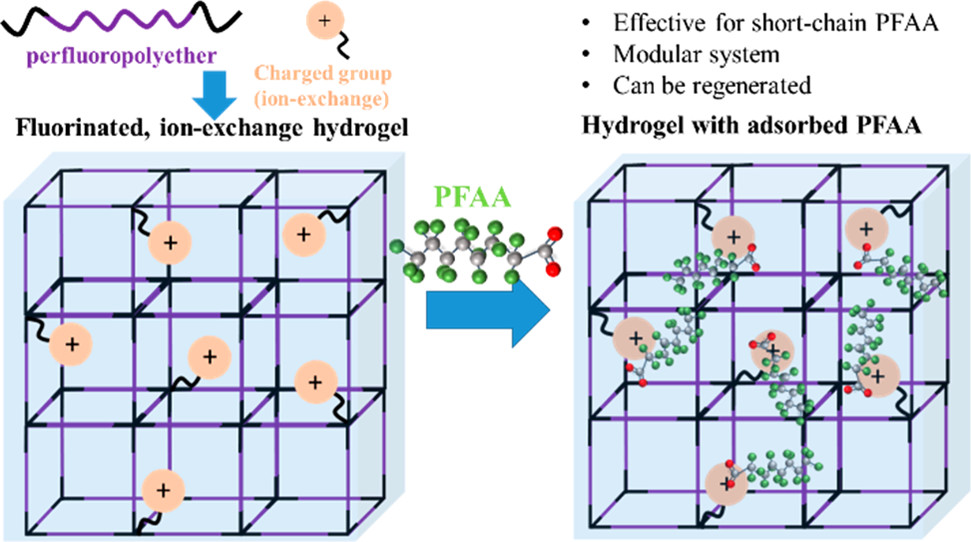
As the old saying (or classic Metallica song) goes, “Fight fire with fire”. When the target is poly- and per-fluoroalkyl substances (PFAS), however, fire will not do the job. PFAS are toxic synthetic chemical contaminants widespread in the environment, including in drinking water and soil. PFAS contain partially fluorinated alkyl tails and can be amphiphilic and lipophilic and dissolve in water, making remediation particularly challenging. There is a need for technologies that can remove and remediate PFAS and perfluoroalkyl acids (PFAA) in the environment to prevent further accumulation and reduce the impacton human health. In this issue of ACS Central Science, Kumarasamy et al. present an innovative strategy for removing PFAS from water with a hydrogel that contains perfluoropolyethers, literally fighting PFAS with PFAS.
143. S. Garcia-Segura, A.B. Nienhauser, A.S. Fajardo, R. Bansal, C.L. Conrad, J.D. Fortner, M. Marcos-Hernández, T. Rogers, D. Villagrán, M.S. Wong, and P. Westerhoff "Disparities between Experimental and Environmental Conditions: Research Steps Towards Making Electrochemical Water Treatment a Reality" Current Opinion in Electrochemistry 22, 9-16 (2020) DOI: 10.1016/j.coelec.2020.03.001
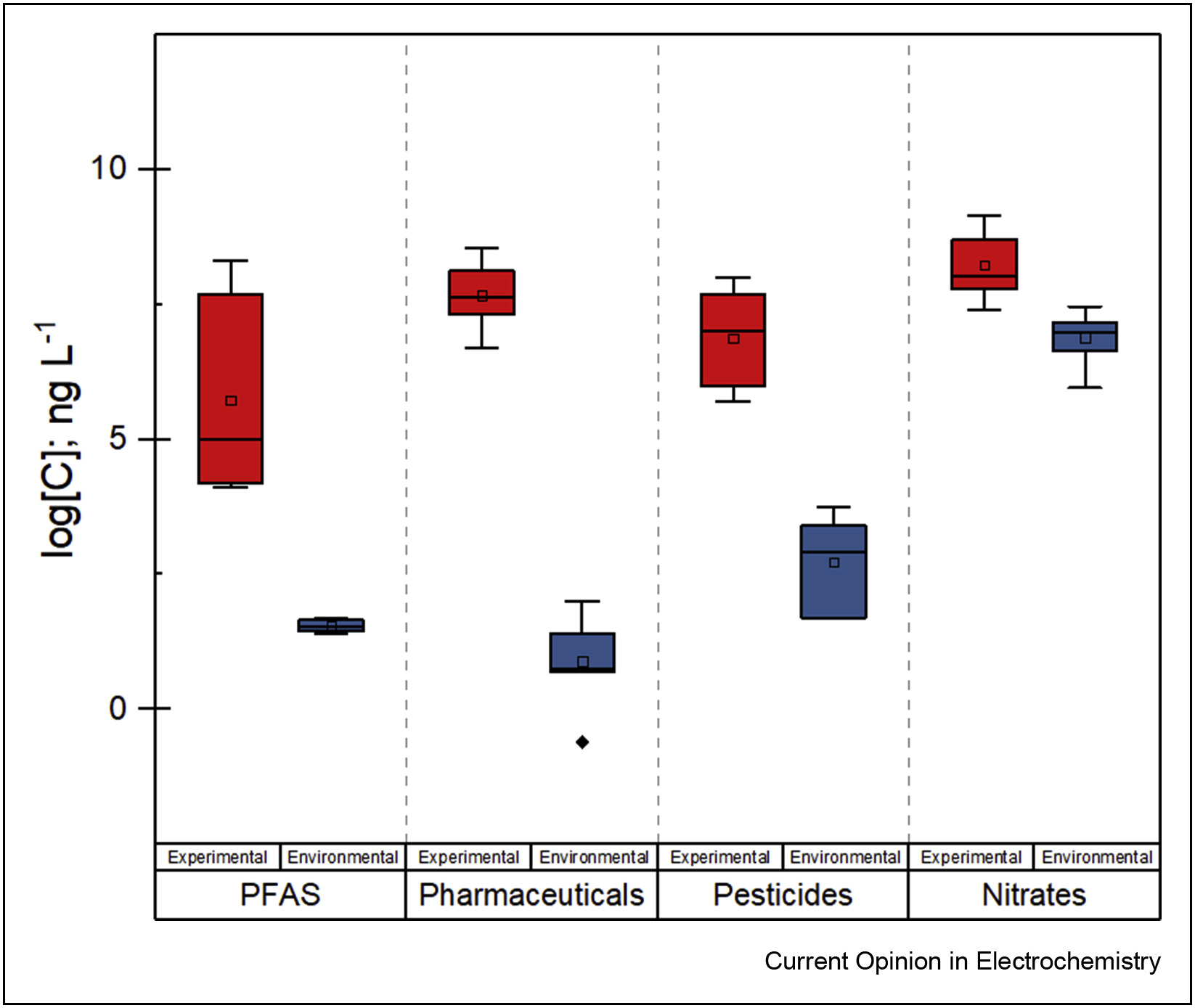
Electrochemical water treatment is one of the key topics of environmental electrochemistry. Identifying electrocatalytic materials capable of electrogenerating high oxidant species in situ seems to have catalyzed researchers’ interest in these processes. While most studies have focused on ideal lab-made solutions, translation to higher technology readiness levels and commercialization requires reframing research questions in context of real water matrices. In this current opinion, we discuss disconnects that may occur when focusing on synthetic solutions treatment rather than real waters. Future research can fill the gaps identified herein, thus facilitating application of electrochemical water treatment technologies.
142. L. Chen, W.C. Elias, Y.B. Yin, Z.C. Zhang, and M.S. Wong "Acid-catalyzed pyrolytic synthesis of levoglucosan through salt-mediated ring locking" Green Chemistry 22, 1968-1977 (2020) DOI: 10.1039/C9GC03973B
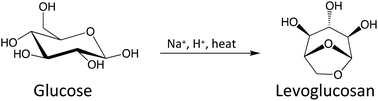
Selectively producing chemicals from cellulosic carbohydrate pyrolysis in large quantities is challenging, especially anhydro-monosaccharides with double-ring, triple-ring, and furan/pyran structures. Formation of these sugar derivatives greatly improves when the pyranose ring opening is inhibited during pyrolysis, which is accomplished by chemically replacing the hydroxyl group at the anomeric carbon with an alkoxy group. A simpler ring-locking approach is required for scalable chemical production, however. In this work, we demonstrate that introducing Na2SO4 and H2SO4 to glucose pyrolysis significantly increases levoglucosan (LGA) formation, from a 6% yield to as high as 40% at 350°C. With H2SO4 as the acid catalyst, Na+ acts to inhibit the ring opening. Glucose pyrolysis with different alkali metal cations (Li+, Na+, K+, Rb+ and Cs+) gives different reaction products, which can be explained largely by an ionic electronegativity effect. Weaker electronegativity promotes the formation of a ring-opened product such as 5-hydroxymethylfurfural (HMF), and stronger electronegativity increases the formation of sequential dehydration products like levoglucosenone (LGO). Sodium has the optimum ionic electronegativity for preferential association with the ring oxygen. The Na2SO4/H2SO4 combination improved LGA yields for all carbohydrate substrates tested (up to 70%), including lignocellulose. These findings highlight the potential of using alkali metal salts to produce anhydrosugars in high yields from cellulosic carbohydrate pyrolysis.
141. C.D. Powell, A.J. Atkinson, Y. Ma, M. Marcos-Hernández, D. Villagran, P. Westerhoff, and M.S. Wong "Magnetic nanoparticle recovery device (MagNERD) enables application of iron oxide nanoparticles for water treatment" Journal of Nanoparticle Research 22, 48 (2020) DOI: 10.1016/j.watres.2020.115540
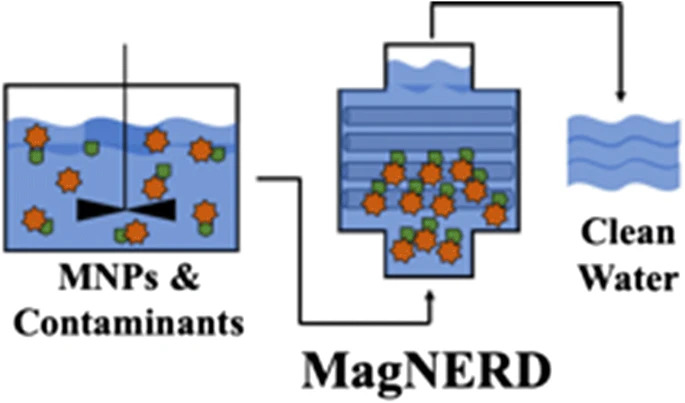
An optimized permanent magnetic nanoparticle recovery device (i.e., the MagNERD) was developed and operated to separate, capture, and reuse superparamagnetic Fe3O4 from treated water in-line under continuous flow conditions. Experimental data and computational modeling demonstrate how the MagNERD’s efficiency to recover nanoparticles depends upon reactor configuration, including the integration of stainless-steel wool around permanent magnets, hydraulic flow conditions, and magnetic NP uptake. The MagNERD efficiently removes Fe3O4 in the form of a nanopowder, up to >95% at high concentrations (500 ppm), under scalable and process-relevant flow rates (1 L/min through a 1.11-L MagNERD reactor), and in varying water matrices (e.g., ultrapure water, brackish water). The captured nanoparticles were recoverable from the device using a simple hydraulic backwashing protocol. Additionally, the MagNERD removed ≥94% of arsenic-bound Fe3O4, after contacting As-containing simulated drinking water with the nanopowder. The MagNERD emerges as an efficient, versatile, and robust system that will enable the use of magnetic nanoparticles in larger scale water treatment applications.
140. Y. Miao, N.W. Johnson, T. Phan, K.N. Heck, P.B. Gedalnga, X. Zheng, D. Adamson, C. Newell, M.S. Wong, and S. Mahendra, "Monitoring, Assessment, and Prediction of Microbial Shifts in Coupled Catalysis and Biodegradation of 1,4-Dioxane and Co-Contaminants" Water Research 173, 115540 (2020) DOI: 10.1016/j.watres.2020.115540
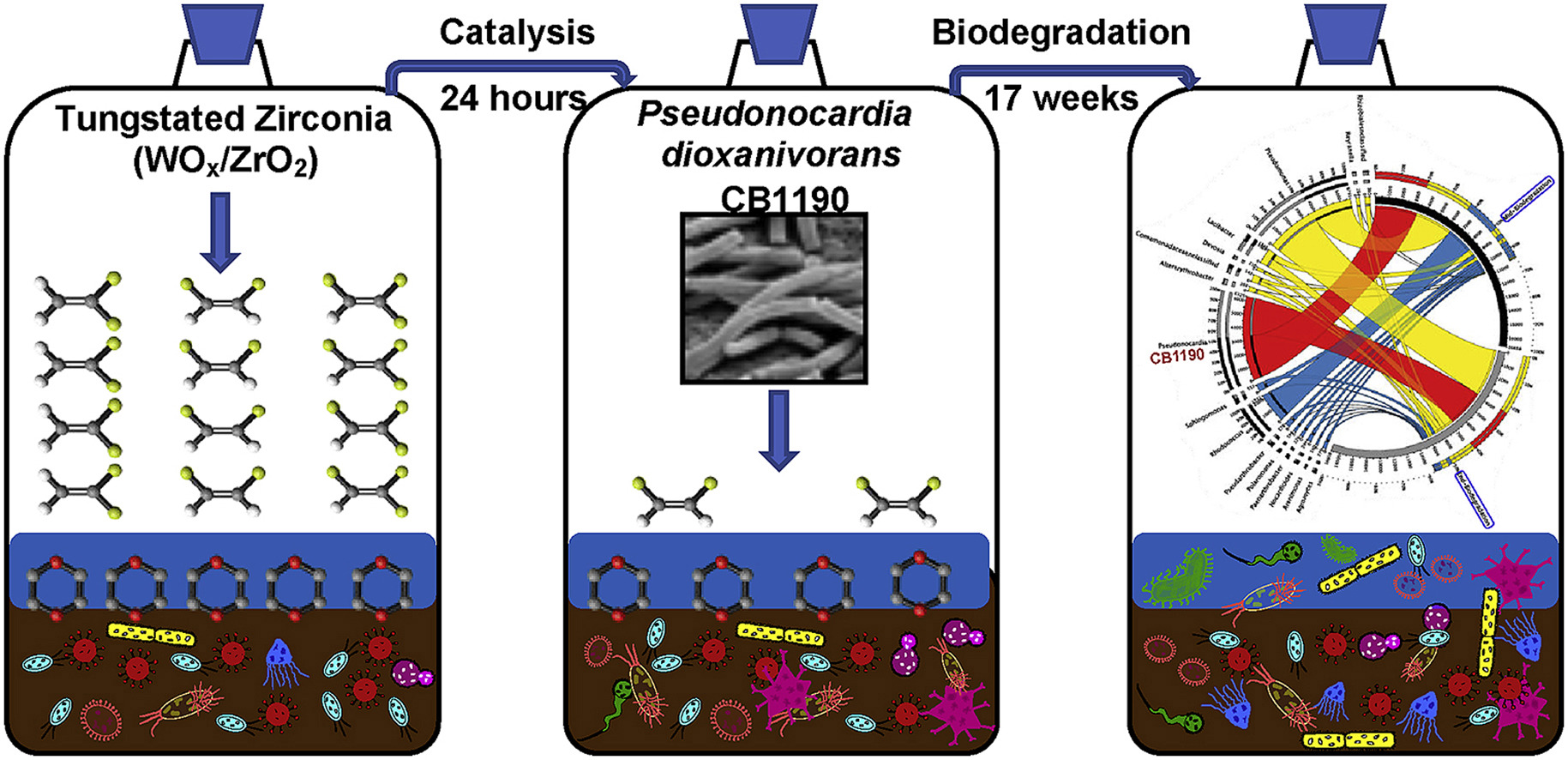
Microbial community dynamics were characterized following combined catalysis and biodegradation treatment trains for mixtures of 1,4-dioxane and chlorinated volatile organic compounds (CVOCs) in laboratory microcosms. Although a few specific bacterial taxa are capable of removing 1,4-dioxane and individual CVOCs, many microorganisms are inhibited when these contaminants are present in mixtures. Chemical catalysis by tungstated zirconia (WOx/ZrO2) and hydrogen peroxide (H2O2) as a non-selective treatment was designed to achieve nearly 20% 1,4-dioxane and over 60% trichloroethene and 50% dichloroethene removals. Post-catalysis, bioaugmentation with 1,4-dioxane metabolizing bacterial strain,Pseudonocardia dioxanivorans CB1190, removed the remaining 1,4-dioxane. The evolution of the microbial community under different conditions was time-dependent but relatively independent of the concentrations of contaminants. The compositions of microbiomes tended to be similar regardless of complex contaminant mixtures during the biodegradation phase, indicating a r-K strategy transition attributed to the shock experienced during catalysis and the subsequent incubation. The originally dominant genera Pseudomonas and Ralstonia were sensitive to catalytic oxidation, and were overwhelmed by Sphingomonas, Rhodococcus, and other catalyst-tolerant microbes, but microbes capable of biodegradation of organics thrived during the incubation. Methane metabolism, chloroalkane-, and chloroalkene degradation pathways appeared to be responsible for CVOC degradation, based on the identifications of haloacetate dehalogenases, 2-haloacid dehalogenases, and cytochrome P450 family. Network analysis highlighted the potential interspecies competition or commensalism, and dynamics of microbiomes during the biodegradation phase that were in line with shifting predominant genera, confirming the deterministic processes guiding the microbial assembly. Collectively, this study demonstrated that catalysis followed by bioaugmentation is an effective treatment for 1,4-dioxane in the presence of high CVOC concentrations, and it enhanced our understanding of microbial ecological impacts resulting from abiotic-biological treatment trains. These results will be valuable for predicting treatment synergies that lead to cost savings and improve remedial outcomes in short-term active remediation as well as long-term changes to the environmental microbial communities.
139. C.L. Conrad, Y.B. Yin, T. Hanna, A.J. Atkinson, P.J.J. Alvarez, T.N. Tekavec, M.A. Reynolds, and M.S. Wong, "Fit-for-Purpose Treatment Goals for Produced Waters in Shale Oil and Gas Fields" Water Research 173, 115467 (2020) DOI: 10.1016/j.watres.2020.115467
Hydraulic fracturing (HF), or “fracking,” is the driving force behind the “shale gas revolution,” completely transforming the United States energy industry over the last two decades. HF requires that 4–6 million gallons per well (15,000–24,000 m3/well) of water be pumped underground to stimulate the release of entrapped hydrocarbons from unconventional (i.e., shale or carbonate) formations. Estimated U.S. production volumes exceed 150 billion gallons/year across the industry from unconventional wells alone and are projected to grow for at least another two decades. Concerns over the environmental impact from accidental or incidental release of produced water from HF wells (“U-PW”), along with evolving regulatory and economic drivers, has spurred great interest in technological innovation to enhance U-PW recycling and reuse. In this review, we analyze U-PW quantity and composition based on the latest U.S. Geographical Survey data, identify key contamination metrics useful in tracking water quality improvement in the context of HF operations, and suggest “fit-for-purpose treatment” to enhance cost-effective regulatory compliance, water recovery/reuse, and resource valorization. Drawing on industrial practice and technoeconomic constraints, we further assess the challenges associated with U-PW treatment for onshore U.S. operations. Presented are opportunities for targeted end-uses of treated U-PW. We highlight emerging technologies that may enhance cost-effective U-PW management as HF activities grow and evolve in the coming decades.
138. C.A. Clark, C.P. Reddy, H. Xu, K.N. Heck, G. Luo, T.P. Senftle, and M.S. Wong, "Mechanistic Insights into pH-Controlled Nitrite Reduction to Ammonia and Hydrazine over Rhodium" ACS Catalysis 10, 494-509 (2020) DOI: 10.1021/acscatal.9b03239
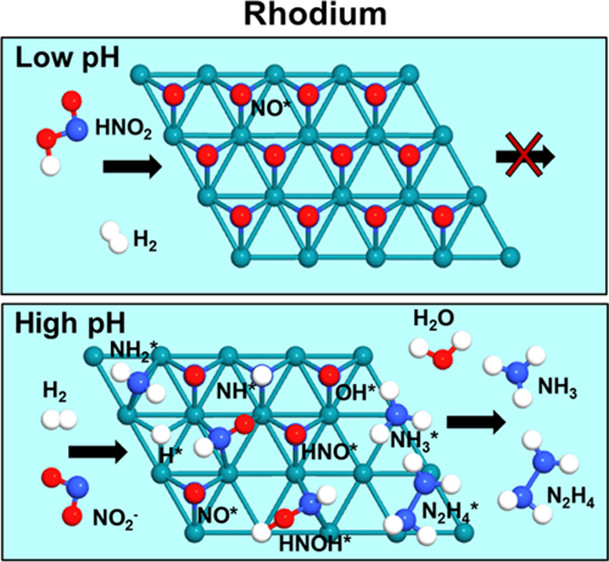
An unintended consequence of industrial nitrogen fixation through the Haber–Bosch process is nitrate (NO3–) and nitrite (NO2–) contamination of ocean, ground, and surface waters from fertilizer runoff. Transition-metal catalysts, particularly those based on Pd, are effective in removing NO3–/NO2– through reduction to N2 or NH4+. Pd is regarded as the most effective metal for NO3–/NO2– reduction, and as such, few studies have thoroughly explored the performance of other transition metals as a function of varying reaction conditions. In this work, we investigated the NO2– reduction properties of alumina-supported Rh using Pd as a benchmark, where we varied the bulk solution pH to probe the effect of reaction conditions on the catalytic chemistry. Pd expectedly showed a high reduction activity (289 L/g-surface-metal/min) and a high N2 selectivity (>99% at 20% conversion) at low pH and near inactivity at high pH. Surprisingly, the Rh catalyst, while inactive at low pH, showed moderate activity (22 L/g-surface-metal/min) and high NH4+ selectivity (>90% at 20% conversion) at high pH. Hydrazine (N2H4) was also detected as a reaction intermediate when NH4+ was formed. Microkinetic models built with energetics from density functional theory reveal that Rh catalysts are poisoned by NO* at low pH because of the rapid dissociative adsorption of protonated nitrite (HNO2) under acidic conditions, which was confirmed by in aqua surface-enhanced Raman spectroscopy. NO* poisoning of the Rh surface lessens at increased solution pH because NO2– does not dissociate as readily compared to HNO2, which explains why Rh exhibits higher activity in basic solutions. The microkinetic models further elucidate the competition between N2H4 and NH3/NH4+ formation as a function of pH, where we find that hydrogen surface coverage dictates product selectivity. These results update the common view that only Pd-based catalysts are effective for NO2– reduction and suggest unexplored avenues for nitrogen chemistry.
137. Y.B. Yin, L. Chen, K.N. Heck, Z.C. Zhang, and M.S. Wong, "Toward glucuronic acid through oxidation of methyl-glucoside using PdAu catalysts" Catalysis Communications 135, 105895 (2020) DOI: 10.1016/j.catcom.2019.105895
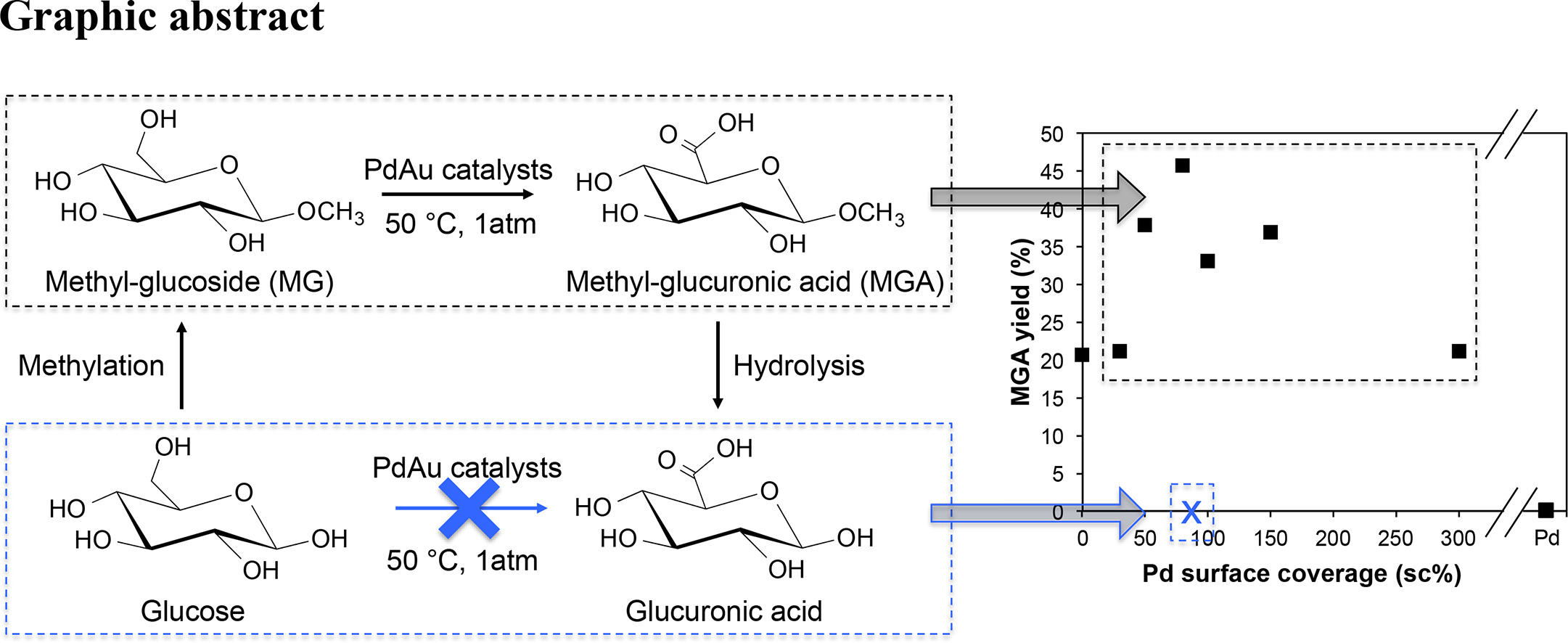
The production of glucuronic acid via enzyme catalysis from biomass is slow. Here we studied the oxidation of methoxy-protected glucose (MG) using Pd-on-Au nanoparticle model catalysts to generate methoxy-protected glucuronic acid (MGA), a precursor to glucuronic acid. Pd-on-Au showed volcano-shape activity dependence on calculated Pd surface coverage (sc). The 80 sc% Pd-on-Au catalyst composition showed maximum initial turnover frequency (413 mol-MG mol-surface-atom−1 h−1) that was 5× higher than that of Au/C, while Pd/C was inactive. This Pd-on-Au composition gave the highest MGA yield (46%), supporting a bimetallic approach to glucuronic acid production.
136. Z. Wang, R. Zhang, Z. Lei, C. Descorme, and M.S. Wong, "New opportunities and challenges in energy and environmental catalysis (EEST2018)" Catalysis Today 339, 1-2 (2020) DOI: 10.1016/j.cattod.2019.08.001

The series of Chinese National Conference on Energy and Environmental Science & Technology (EEST) found its origin in 2016 in Shenyang, China. The aim of the conference is to provide a venue for scientists from both academia and industry to discuss the status and prospects of energy and environmental science & technology. With the theme of “the opportunities and challenges in energy and environmental science”, EEST2018 was held during April 6–9, 2018, in Beijing, China. This conference was co-organized by the China Energy Society and Beijing University of Chemical Technology. The scientific program consisted of six plenary lectures, 34 keynote lectures, 113 oral presentations and 170 poster presentations. Plenary lectures were delivered by Prof. S. Ted Oyama (The University of Tokyo, Virginia Polytechnic Institute), Prof. In-Sik Nam (Pohang University of Science and Technology), Prof. Qiang Xu (National Institute of Advanced Industrial Science and Technology), Prof. Hui-Ming Chen (Institute of Metal Research, Chinese Academy of Sciences), Prof. Tianwei Tan (Beijing University of Chemical Technology) and Prof. Hong He (Research Center for Eco-Environmental Sciences, Chinese Academy of Sciences).
135. Y.B. Yin, K.N. Heck, C.L. Conrad, C.D. Powell, S. Guo, M.A. Reynolds, and M.S. Wong, "PdAu-catalyzed oxidation through in situ generated H2O2 in simulated produced water" Catalysis Today 339, 362-370 (2020) DOI:10.1016/j.cattod.2019.05.001
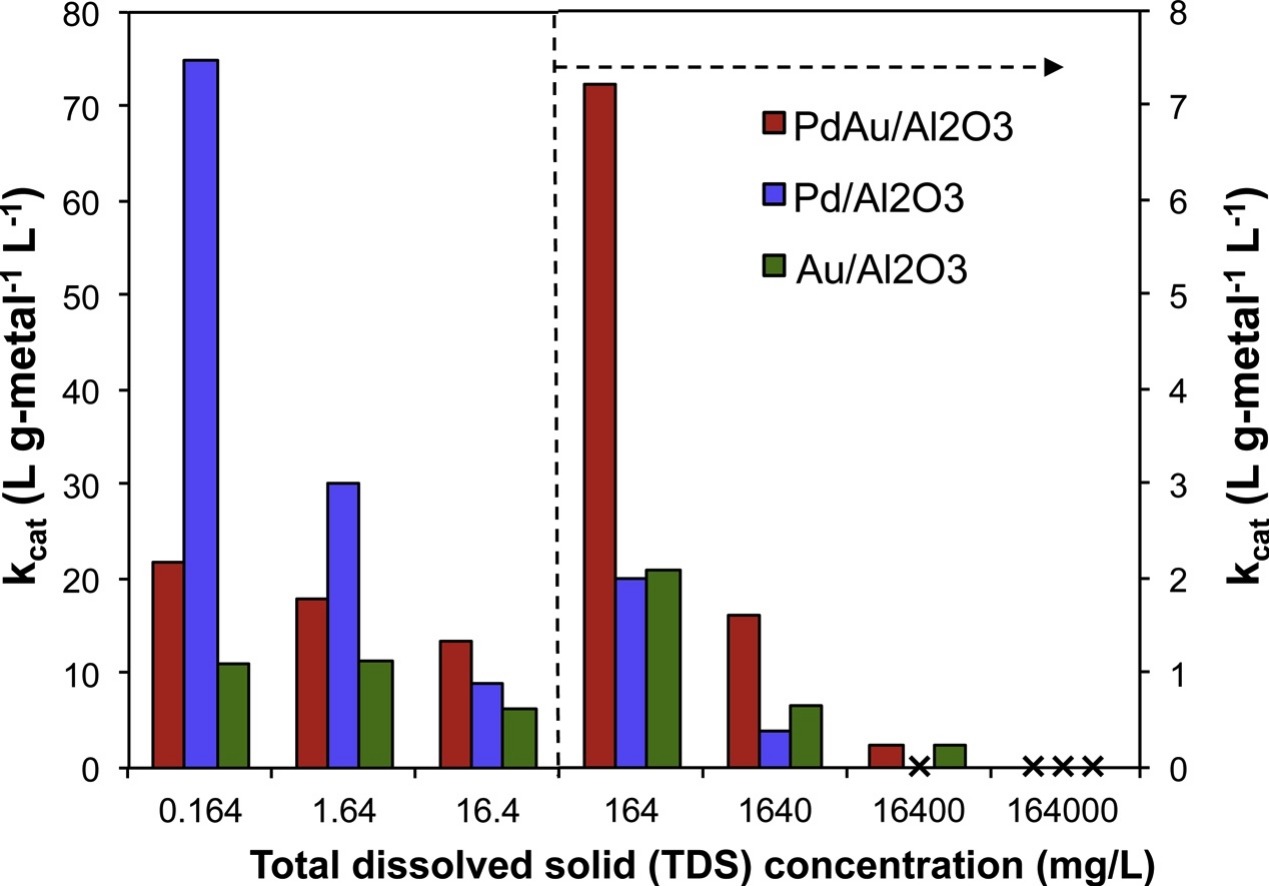
Most wastewater recovered from hydraulically fractured oil and gas wells (i.e. produced water) is transported to government-permitted, salt-water disposal units (SWDs) and discarded via downhole injection. However, there is a limited availability of disposal wells in some states and growing interest over future options for beneficial reuse. One alternative to using SWD facilities is to recycle the water for further use in oilfield operations. Residual oil and grease are one contaminant class in produced water where cost-effective treatment technologies are lacking. In this work, we studied the ability of alumina-supported bimetallic PdAu to degrade organic compounds at room temperature and atmospheric pressure via the catalytic formation of H2O2. Similar to monometallic Pd and Au catalysts, the PdAu catalyst produced H2O2 and hydroxyl radicals in the presence of oxygen and formic acid. The bimetallic catalyst was the most active in terms of initial OH formation rate, and when phenol was present, PdAu showed the highest rate of phenol degradation. We assessed the promotional and inhibitory effects of other species present in produced water including ferrous ion concentration, pH and salt concentration on catalytic phenol oxidation. PdAu was catalytically active for phenol degradation in simulated produced water at salinities as high as ˜0.3 M (˜16,000 ppm). The combination of air-formic acid-bimetallic catalyst is an intriguing approach for the degradation of organics in contaminated water at low pH and moderate salinity.