2013 Abstracts.
100. J. C. Velázquez, S. Leekumjorn, Q. X. Nguyen, Y. L. Fang, K. N. Heck, G. D. Hopkins, M. Reinhard, M. S. Wong, "Chloroform Hydrodechlorination Behavior of Alumina-supported Pd and PdAu Catalysts" AIChE Journal, 59(12), 4474–4482 (2013). DOI: 10.1002/aic.14250
Chloroform is a common groundwater contaminant that is very difficult to remove. Chemically converting it into a less toxic form through heterogeneous catalysis is an attractive approach over conventional physical removal methods if it can be done economically. In this study, we explore the efficacy of supported precious metal catalysts for chloroform hydrodechlorination. We find that Pd/Al2 O3 is catalytically active for this reaction (6.4 L/gPd/min) at room temperature, atmospheric pressure, in buffered water, and in the presence of hydrogen gas, and that Pd deposited on commercial Au/Al2O3 shows activities as high as 22.4 L/gPd/min, suggestive of some Pd metal located on top of Au domains. The primary reaction product is methane, with selectivity values exceeding 90%. Surface-enhanced Raman spectroscopy shows evidence of chloroform adsorption and dechlorination on the catalyst surface under aqueous conditions. The results highlight the potential of ambient-condition reductive catalysis to remove chloroform from water.
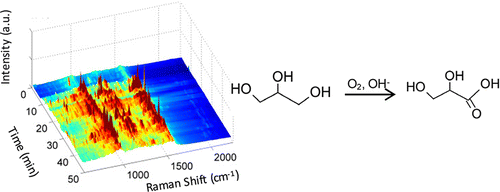
99. K. N. Heck, B. G. Janesko, G. E. Scuseria, N. J. Halas, M. S. Wong, "Using Catalytic and Surface-Enhanced Raman Spectroscopy-Active Gold Nanoshells to Understand the Role of Basicity in Glycerol Oxidation" ACS Catalysis, 3, 2430–2435 (2013). DOI: 10.1021/cs400643f
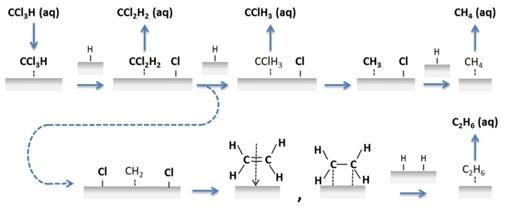
The origin of oxidation activity of gold catalysts has been a subject of great interest, particularly with the discovery of selective glycerol oxidation under water-phase alkaline conditions, for which neither small gold nanoparticles nor a catalyst support is necessary for activity. Little is known about the interactions among the catalyst surface, reactant, and hydroxyl species, which have never been examined spectroscopically because of a lack of developed in situ methods. In this work, we studied the room-temperature, water-phase reaction of glycerol oxidation using gold nanoshells (Au NSs), in which the gold substrate was active for surface-enhanced Raman spectroscopy (SERS) and catalysis. Analysis of glycerol solutions at high pH values and with oxygen content indicated that glycerol and glycerolate species did not bind directly to the catalyst surface in the absence of oxygen. However, glycerate surface species formed very rapidly when oxygen was present, suggesting an Eley–Rideal-type reaction mechanism with O2 (and/or O2-activated OH–) as the adsorbed species. SERS analysis of carbon monoxide chemisorption on Au NSs indicated that higher pH values progressively weakened the C–O bond as the Au negative charge increased. The importance of high alkalinity to Au-catalyzed alcohol oxidation may result from both the activation of glycerol via deprotonation and the weakening of the adsorbed O2 double bond via induced Au negative charge.
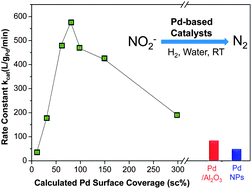
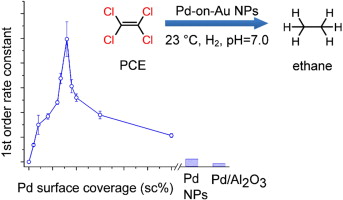
98. Z. Zhao, Y.-L. Fang, P. J.J. Alvarez, M. S. Wong, "Degrading perchloroethene at ambient conditions using Pd and Pd-on-Au reduction catalysts," Appl. Catal. B-Environ., 140-141, 468-477 (2013). DOI: 10.1016/j.apcatb.2013.04.032
Perchloroethene (PCE) is a common groundwater contaminant, due to its common use as a dry-cleaning solvent. Current treatment methods are limited in their ability to remove PCE from contaminated sites in an efficient and cost effective manner. Palladium-on-gold nanoparticles (Pd-on-Au NPs) have been shown to be highly catalytically active in the hydrodechlorination (HDC) of trichloroethene (TCE) and other chlorinated compounds. However, the catalytic chemistry of such nanoparticles for PCE HDC in water has not been systematically addressed in the literature. In this paper, we assess the catalytic properties of ~4 nm Pd-on-Au NPs, ~4 nm Pd NPs, and Pd/Al2O3 for water-phase PCE HDC under ambient conditions. The Pd-on-Au NPs exhibited volcano-shape activity as a function of Pd surface coverage (sc). Maximum activity was at 80 ± 0.8 sc% (pseudo-first order rate constant of ~5000 L/gPd/min), which was ~20× and ~80× higher than that for Pd NPs and Pd/Al2O3 at room temperature and pH 7. A complete mechanistic model of PCE HDC that coupled gas–liquid mass transfer with the surface reactions was developed and found to be consistent with the observed concentration-time profiles for the 3 catalyst types. The formation and subsequent reaction of daughter products (TCE, dichloroethene isomers, vinyl chloride, and ethene) followed the stepwise dechlorination of the PCE chlorine groups. The final reaction products were ethane and minor amounts of n-butane/butenes. This study establishes the enhanced degradation chemistry of PCE using model Pd-on-Au catalysts and suggests the volcano-shape structure-activity dependence can be generalized from PCE and TCE to other organohalides.
97. S. S. Warudkar, K. R. Cox, M. S. Wong, G. J. Hirasaki, "Influence of stripper operating parameters on the performance of amine absorption systems for post-combustion carbon capture: Part II. Vacuum strippers," Int. J. Greenh. Gas. Con. 16, 351-360, (2013). DOI: 10.1016/j.ijggc.2013.01.049
The alkanolamine absorption process is viewed favorably for use in the separation of carbon dioxide (CO2) from point emission sources such as coal-fired power plants. At present, natural gas sweetening is the most important application for this technology. However, on a number of accounts such as the feed conditions of gas, its composition and process economics; natural gas sweetening and carbon capture are very different applications. Current technology is optimized toward providing a high performance for the former. As a part of this two-part study, we have used the process simulation software ProMax® to perform a detailed analysis on the effect of stripper operating pressure on factors like reboiler energy duty, absorber and stripper column sizing and parasitic power loss. We have examined the performance of monoethanolamine (MEA), diethanolamine (DEA) and diglycolamine (DGA) which are all commercial absorbents that can be reliably modeled in ProMax®. In part I of this study, we have analyzed the performance of strippers operated at pressures ranging from 150 kPa to 300 kPa. In this part of the study, we examine the performance of vacuum strippers operating under low vacuum at pressures of 30 kPa, 50 kPa and 75 kPa. Since vacuum strippers operate at lower temperatures than conventional stripper configurations, it is possible to use waste heat in the reboiler. In this study, we explore this possibility and consider 5 scenarios in which varying fractions of the reboiler steam are provided from waste heat sources located outside the turbine system. As with the cases presented in Part I, our comparisons of different configurations are based on energy consumption and column dimensions required for 90% CO2 capture (separation + compression) from a 400 MW coal-fired power plant. CO2 separated from the flue gas is compressed to a pressure of 16 MPa, typically maintained in the pipelines. On the basis of our findings, we report that vacuum stripping is an attractive altrnative to conventional stripping. It is particularly attractive if significant sources of waste heat outside the turbine system can be located. We also conclude from our work that DEA and DGA have a superior performance than MEA when vacuum strippers are used. Use of vacuum strippers will certainly result in increased capital costs due to the need for larger equipment. However, in the view of potential savings in operating costs mainly by reduction in parasitic power loss; the increased capital expenditure may be justifiable.
96. S. S. Warudkar, K. R. Cox, M. S. Wong, G. J. Hirasaki, "Influence of stripper operating parameters on the performance of amine absorption systems for post-combustion carbon capture: Part I. High pressure strippers," Int. J. Greenh. Gas. Con. 16, 342-350, (2013). DOI: 10.1016/j.ijggc.2013.01.050
Amine absorption is a mature technology that is widely applied on a commercial scale for the removal of acid gases from gas mixtures. It is viewed as the most promising technology to separate carbon dioxide (CO2) at point emission sources such as fossil fuel fired power plants as a part of a strategy called ‘carbon capture and storage’ being deployed to mitigate climate change. However, there are major challenges in advancing its use for this application; the most prominent one being – cost. It is estimated that the application of current technology to CO2 capture will result in a 70–100% increase in the cost of electricity (COE). As a part of this two-part study, we have examined the influence of stripper operating parameters on carbon capture at a 400 MW pulverized coal-fired power plant retrofitted with amine absorption technology. We use the process simulation software, ProMax® to simulate the amine absorption process. Three commercial absorbents namely monoethanolamine (MEA), diethanolamine (DEA) and diglycolamine (DGA) are considered in this work. All the absorbents examined are studied at the typical working concentrations used in commercial operation. We also constrain the CO2 loading of rich amine solution to 0.4 mol-CO2/mol-amine to closely resemble the chemical environment in commercial operation. The main pointer used to compare system performance is the parasitic power loss due to the carbon capture. Parasitic power loss comprises of the loss of plant output due to withdrawal of reboiler steam from the low pressure turbine; the electricity required for CO2 compression and by the plant auxiliaries such as blowers and pumps. We also examine the absorber and stripper column sizes and heat exchanger parameters which are key components of the system. On the basis of our findings in the first part of this study, we report that operating the stripper at higher pressures has significant advantages but also some disadvantages and operational challenges. We also report that DEA and DGA have a superior performance to MEA when used in high pressure strippers. We conclude that increasing the stripper pressure reduces the parasitic losses and the equipment sizing but may result in higher solvent losses and equipment corrosion.
95. H. Qian, L. A. Pretzer, J. C. Velazquez, Z. Zhao, and M. S. Wong, "Gold nanoparticles for cleaning contaminated water," J. Chem. Tech. Biotech. (online). DOI: 10.1002/jctb.4030
Pollutants in the form of heavy metals, fertilizers, detergents, and pesticides have seriously reduced the supply of pure drinking water and usable water. Gold metal has intriguing potential to deal with the water pollution problem, as recent research on several fronts is advancing the concept of nanoscale gold as the basis for cost-effective nanotechnology-based water treatment. Nano-gold has special properties, such as enhanced catalytic activity, visible surface plasmon resonance color changes, and chemical stability, that make it more useful than other materials. This Perspective article highlights the current use of gold nanoparticles for the efficient removal and the selective and sensitive detection of a variety of pollutants in water. The challenges in further developing nano-gold to address water contamination are discussed, which should stimulate future research into improved removal and detection of undesirable chemical compounds.
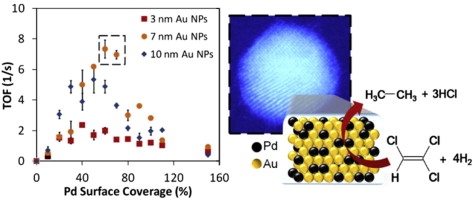
94. L. A. Pretzer, H. J. Song, Y. L. Fang, Z. Zhao, N. Guo, T. Wu, I. Arslan, J. T. Miller, and M. S. Wong, "Hydrodechlorination catalysis of Pd-on-Au nanoparticles varies with particle size," J. Catal., 298, 206-217 (2013). DOI: 10.1016/j.jcat.2012.11.005
Trichloroethene (TCE), a common carcinogen and groundwater contaminant in industrialized nations, can be catalytically degraded by Au nanoparticles partially coated with Pd (“Pd-on-Au NPs”). In this work, we synthesized Pd-on-Au NPs using 3, 7, and 10 nm Au NPs with Pd surface coverages between 0–150% and studied how particle size and composition influenced their TCE hydrodechlorination (HDC) activity. We observed volcano-shape dependence on both Au particle size and Pd surface coverage, with 7 nm Au NPs with Pd coverages of 60–70% having maximum activity. Using extended X-ray absorption fine-structure spectroscopy, we found a strong correlation between catalytic activity and the presence of 2-D Pd ensembles (as small as 2–3 atoms). Aberration-corrected scanning transmission electron microscopy further confirmed the presence of Pd ensembles. The Pd dispersion and oxidation state generally changed from isolated, metallic Pd atoms to metallic 2-D Pd ensembles of varying sizes, and to partially oxidized 3-D Pd ensembles, as Pd surface coverage increased. These changes occurred at different surface coverages for different Au particle sizes. These findings highlight the importance of controlling particle size and surface coverage in bimetallic catalysts.
93. N. Soultanidis, and M. S. Wong, "Olefin impurity effect on n-pentane bimolecular isomerization over WOx/ZrO2," Catal. Commun., 32, 5-10 (2013). DOI: 10.1016/j.catcom.2012.11.017
Depending on the catalyst used, the reaction conditions and the molecules converted, alkane isomerization proceeds via a monomolecular or a bimolecular mechanism. n-Pentane isomerizes via both mechanisms which makes it an excellent probe molecule. In this study, n-pentane isomerization was investigated using WOx/ZrO2. Propylene and 1-pentene were co-fed separately to study the effect on n-pentane isomerization and it was identified that only 1-pentene increases the isomerization selectivity to isopentane. Our results support the model of a bimolecular mechanism that proceeds via a C10 intermediate on Zr-WOx Brønsted sites.
92. M. D. Blankschien, L. A. Pretzer, R. Huschka, N. J. Halas, R. Gonzalez, and M. S. Wong, "Light-triggered biocatalysis using thermophilic enzyme–gold nanoparticle complexes," ACS Nano, 7 (1), 654-663 (2013). DOI: 10.1021/nn3048445
The use of plasmonic nanoparticle complexes for biomedical applications such as imaging, gene therapy, and cancer treatment is a rapidly emerging field expected to significantly improve conventional medical practices. In contrast, the use of these types of nanoparticles to noninvasively trigger biochemical pathways has been largely unexplored. Here we report the light-induced activation of the thermophilic enzyme Aeropyrum pernix glucokinase, a key enzyme for the decomposition of glucose via the glycolysis pathway, increasing its rate of reaction 60% with light by conjugating the enzyme onto Au nanorods. The observed increase in enzyme activity corresponded to a local temperature increase within a calcium alginate encapsulate of 20°C when compared to the bulk medium maintained at standard, nonthermophilic temperatures. The encapsulated nanocomplexes were reusable and stable for several days, making them potentially useful in industrial applications. This approach could significantly improve how biochemical pathways are controlled for in in vitro and, quite possibly, in vivo use.