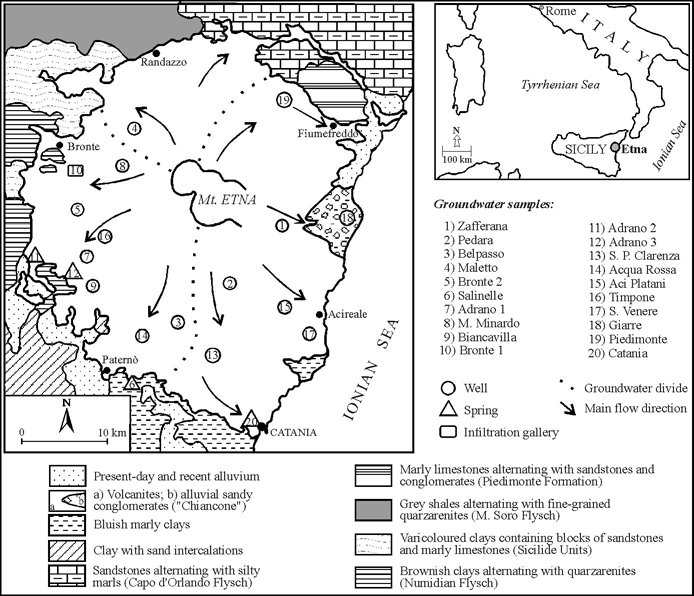
Figure 1. Generalized hydrogeologic map of Mt. Etna showing the distribution of principal lithologic units and our sample locations.
by
M. Pennisi1, W.P. Leeman2,
S. Tonarini1, A. Pennisi3, P.I.
Nabelek4
1 Istituto di Geocronologia e Geochimica Isotopica, Via Maffi 36, 56125, Pisa, Italy
2 Keith-Wiess Geological Laboratories, Rice University, Houston TX 77005, USA
3 S.IDR.A. Municipal Aqueduct Enterprise of Catania (Italy) Catania, Italy
4 Department of Geological Sciences, University of Missouri, Columbia MO 65211, USA
(in press, Geochimica et Cosmochemica Acta)
Abstract - Combined B, O, H, and Sr
isotopic studies of groundwaters from Mt. Etna provide new
constraints on their origin and the consequences of fluid-rock
interaction within this hydrologic system. Variations in
d18O
(-9.7 to -7.2‰) and dD
(-62 to -23‰) mainly lie along the regional meteoric water line
and suggest that most waters originated as local precipitation.
However, small shifts in d18O,
and variable 87Sr/86Sr (0.70355 to 0.70879) and
d11B
(-5.2 to +25.8‰) indicate that subsequent interactions occurred
between the fluids and local rocks. High B/Cl ratios in all samples
seemingly preclude direct involvement of seawater in the hydrologic
system despite the proximity to the coast and, in some samples,
elevated 87Sr/86Sr and
d11B.
Two general 'end-member' water types are recognized on the basis of
their chemistry. These apparently are produced by interactions of
local meteoric waters with the dominant reservoir rocks - either
basaltic lavas of Etna or the underlying sediments; high
d11B
in the sediment-hosted end-member points to a significant marine
carbonate contribution. Subsequent mixing between these or similar
end-members produced a range of intermediate composition
groundwaters. Certain anomalous water compositions require the
presence of a distinct component with high B and moderate
d11B
(ca. +10‰) but relatively low 87Sr/86Sr;
an anthropogenic source for this component is plausible. One unusual
sample has B and Sr isotopic compositions similar to the other
volcanic rock-hosted waters, but anomalously high Cl content that
likely reflects local magmatic outgassing near this sampling
locality. In general, this study indicates that groundwater B and Sr
isotopic compositions are rock-dominated; these data provide valuable
constraints on the origin and evolution of groundwaters.
Figures from this paper are reproduced below:
|
|
|
Figure 1. Generalized hydrogeologic map of Mt. Etna showing the distribution of principal lithologic units and our sample locations. |
|
|
Figure 2. Hydrogen and oxygen isotopic composition of Etna ground-waters. Meteoric water lines for the central Mediterranean sea (dD = 8 • d18O +18; MMW/A = Anzá et al., 1989) and for southern Italy (dD = 8 • d18O +22; MMW/P = Panichi and Noto, 1992) are shown for reference; GMW represents the global meteoric water trend of Craig (1961). A field for pore waters in ocean sediments (Lawrence and Gieskes, 1981) indicates the compositional changes in seawater as a result of variable degrees of reaction (increasing to the left) with oceanic crustal rocks. Similar effects could explain the unusual compositions of samples AR and ACI. In this and subsequent figures, samples are identified as follows: volcanic-hosted (filled circles), sediment-hosted (open squares), 'indeterminant host' (filled triangles). Key to distinctive analyses: CR = S.P. Clarenza, AR = Acqua Rossa, ACI = Aci Platani. |
|
|
Figure 3. Boron vs. chlorine plot for Etna groundwaters. All samples exhibit high B/Cl with respect to seawater (SW; B/Cl= 0.00075). The waters of S.P. Clarenza (CR) and Acqua Rossa (AR) are notable with respect to their unusually high B/Cl values. Key to other points of interest: SV = S. Venere, SAL = Salinelle, ZF = Zafferana; EM-1 and EM-2 (small open circles) are hypothetical end-members that bracket the trend defined by most Etna waters (shaded area). Lines are shown representing simple mixing between EM-1 and EM-2, SV, SW, and CR (these are curved because of the log axes). |
|
|
Figure 4. (a) d11B vs. B/Cl, and (b) 87Sr/86Sr vs. B/Cl diagrams. Both panels show an inverse correlation for most samples that can be approximated by two-component mixing relations between EM-1 and either EM-2 or SV end-members. Mixing lines between EM-1 and seawater (SW) show poor correspondence to Etna water compositions. S.P. Clarenza (CR) and Acqua Rossa (AR) waters are anomalous with respect to their excessive B content and elevated d11B. The high-Cl Zafferana water (ZF) cannot be represented by mixing of any known end-member combinations, and likely contains excess Cl from local magmatic outgassing. |
|
|
Figure 5. (a) d11B vs. B, and (b) 87Sr/86Sr vs. Sr diagrams illustrating possible relations between volcanic rock- and sediment-hosted hydrologic reservoirs and representative external fluid sources. Literature boron isotopic data are shown for carbonate (+; larger symbols represent modern biogenic carbonates) and siliciclastic (x) sediments (see text for references). Calculated curves are shown for mixing between EM-1 and EM-2, SV, SW, CR, and a hypothetical meteoric water (MW; small x's); end member compositions are given in Table 3. d11B = +35‰; 0.01 ppm B). Heavy lines show effects of MW reaction with either Etna basalts, pelitic sediment, or carbonates as calculated using the model of Banner and Hanson (1990); in panel (b) the latter two curves overlap. Average compositions were used in modeling for flysch (200 ppm Sr, 100 ppm B, d11B = -5‰, 87Sr/86Sr = 0.70815) and carbonate sediments (1000 ppm Sr, 43 ppm B, d11B = +23‰, 87Sr/86Sr = 0.70815); note that the isotopic compositions adopted for carbonates represent minimal values needed to model the EM-1 or SV end members. |
|
|
Figure 6. d11B vs. 87Sr/86Sr plot showing possible mixing relations between EM-1 and EM-2, SV, CR, or SW; most 'type-b' and 'type-c' samples are bracketed by the mixing curves. Heavy curves indicate progressive reaction of meteoric water (MW) with basaltic (small open circles) and sedimentary host rocks (see text); separate curves are shown for flysch (small open squares) and carbonate (small filles squares) reactants. These curves suggest that EM-1 is similar to MW that has reacted strongly with Etna lavas, whereas EM-2 (or similar variants) could represent MW that has reacted extensively with sediments. Note that, by assuming moderately higher 87Sr/86Sr values for the sediments, it is easy to produce a spectrum of end members bracketing SV and EM-2. Comparable reaction paths for water-rock interaction with basalts and sediments are produced by model of Banner and Hanson (1990); for clarity these are not shown. |
|
|
Figure 7. (a) d11B vs. d18O, and (b) 87Sr/86Sr vs. d18O diagrams for Etna waters. Note that d18O data are lacking for several samples. Curves are shown for simple mixing between EM-1 and EM-2, SW, and MW (small x's). Heavy reaction lines show the effects of progressive equilibration of MW with basaltic (small open circles) and for carbonate (small filled squares) and flysch (small open squares) sediments. Banner and Hanson (1990) water-rock interaction paths virtually overlap the equilibrium curves and, for clarity, are not shown. As in Fig. 6, these curves pass near the positions of the principal water end-members (EM-1, EM-2) and are consistent with an initial meteoric origin of each. Open boxes around MW cpmpositions reflect the range in d18O seen in Fig. 2. |
|
|
Figure 8. (a) d11B vs. dD, and (b) 87Sr/86Sr vs. dD diagrams for Etna waters. Note that dD data are lacking for several samples. Curves are shown for simple mixing between EM-1 and EM-2, SW, and MW (small x's). Heavy reaction lines show the effects of progressive equilibration of MW with basaltic (small open circles) and either carbonate (small filled squares) or flysch (small open squares) reactants; again Banner-Hanson reaction paths are virtually identical. Open boxes around MW cpmpositions reflect the range in dD seen in Fig. 2. |
|
|