2022 Abstracts.
177. L. Duan, B. Wang, K.N. Heck, C.A. Clark, J. Wei, M. Wang, J. Metz, G. Wu, A. Tsai, S. Guo, J. Arrendondo, A.D. Mohite, T.P. Senftle, P. Westerhoff, P.J.J. Alvarez, X. Wen, Y. Song and M.S. Wong "Titanium oxide improves boron nitride photocatalytic degradation of perfluorooctanoic acid" Chemical Engineering Journal 448, 137735 (2022) DOI: 10.1016/j.cej.2022.137735
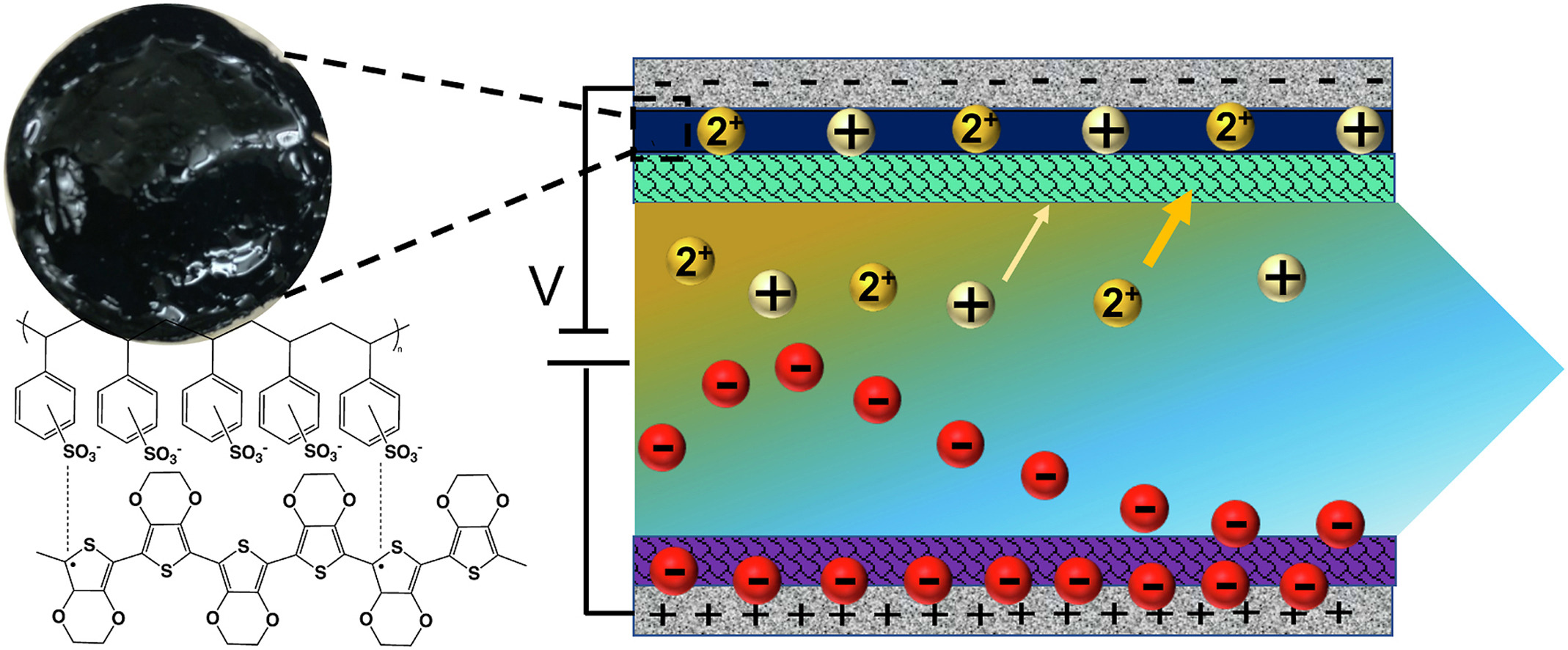
There is a need for membranes and processes that can selectively separate target ions from other similar ionic species. Recent studies have shown that electrified processes for ion removal such as membrane capacitive deionization (MCDI) and electrodialysis (ED) are selective towards specific ionic species, but selectivities are generally limited. Here, we demonstrate that an ion-selective polymer coating can significantly enhance ion selectivities for MCDI processes. We focused on the preferential removal of Ca2+ over Na+ and used the conductive and sulfonated polymer poly(3,4-ethylene dioxythiophene):polystyrene sulfonate (PEDOT:PSS) as a model selective ion-exchange coating. We first measured the permeability of Ca2+ and Na+ in freestanding PEDOT:PSS membranes of varying crosslink density and found that the permeability of Ca2+ was six times greater than that for Na+ in optimized membranes. Next, we used PEDOT:PSS in an MCDI process by depositing thin PEDOT:PSS coatings on top of composite electrodes. We found that the PEDOT:PSS coatings significantly enhanced the preferential permeability of Ca2+ over Na+ relative to unmodified electrodes and produced a preferential removal as high as 8:1 on a molar basis. This work demonstrates a new approach to enhance selective ion removal in MCDI and other electro-driven ion separation processes.
176. J. Metz, P. Zuo, B. Wang, M.S. Wong, and P.J.J. Alvarez "Perfluorooctanoic acid Degradation by UV/Chlorine" Environmental Science & Technology Letters 9, 8, 673-679 (2022) DOI: 10.1021/acs.estlett.2c00452
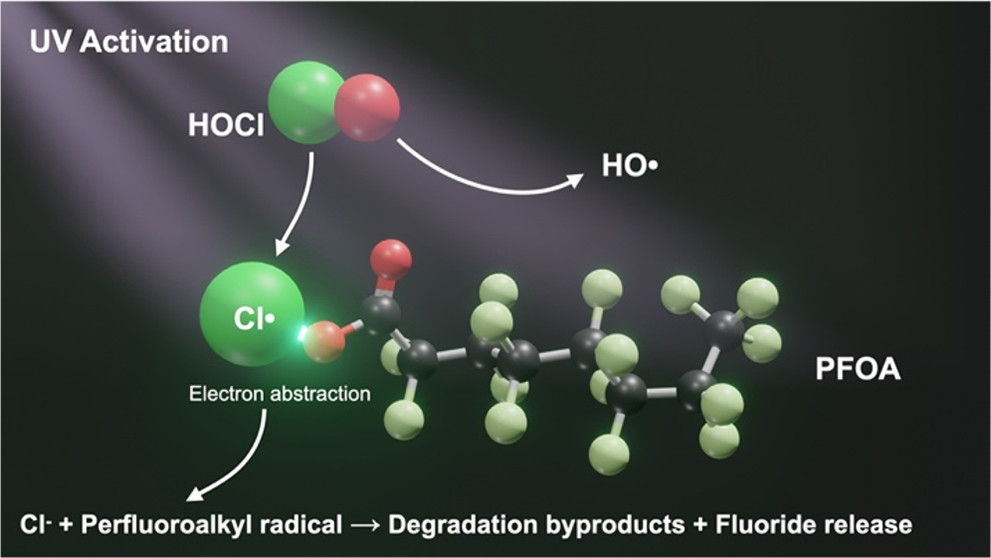
While per- and polyfluoroalkyl substances (PFAS) are recalcitrant to chemical reactions traditionally used in water treatment, we report the novel finding that combining ultraviolet (UV, 254 nm) light and chlorine can promote perfluorooctanoic acid (PFOA) degradation. About 12% removal of 100 μg/L PFOA was observed after 30 min of irradiation (6.5 × 10–6 Einstein L–1 s–1) in the presence of 1.4 mM (106 mg/L) NaOCl, compared to only 1% removal by UV photolysis and no removal by NaOCl alone. UV/chlorine with 0.02 mM NaOCl (1.5 mg/L, a more common dose for water treatment) removed 6 μg/L PFOA within 30 min. To better detect defluorination, 50 mg/L PFOA was used, and UV/chlorine released significantly more fluoride (382 μg/L) than UV photolysis (0 μg/L) and dark controls (0 μg/L) over 30 min. By 60 min, this represents 32% of the maximum possible defluorination for the amount of PFOA removed by UV/chlorine versus 2% for UV photolysis. Radical scavenger tests indicated that Cl• and Cl2•– play a crucial role in PFOA degradation, which we postulate is initiated by electron abstraction leading to a decarboxylation–hydroxylation–elimination–hydrolysis pathway. Whereas reaction rates were relatively slow for practical application in water treatment plants, these results underscore overlooked reactions with common water treatment constituents that may influence the fate of PFAS.
175. L. Duan, B. Wang, K.N. Heck, C.A. Clark, J. Wei, M. Wang, J. Metz, G. Wu, A. Tsai, S. Guo, J. Arrendondo, A.D. Mohite, T.P. Senftle, P. Westerhoff, P.J.J. Alvarez, X. Wen, Y. Song and M.S. Wong "Titanium oxide improves boron nitride photocatalytic degradation of perfluorooctanoic acid" Chemical Engineering Journal 448, 137735 (2022) DOI: 10.1016/j.cej.2022.137735
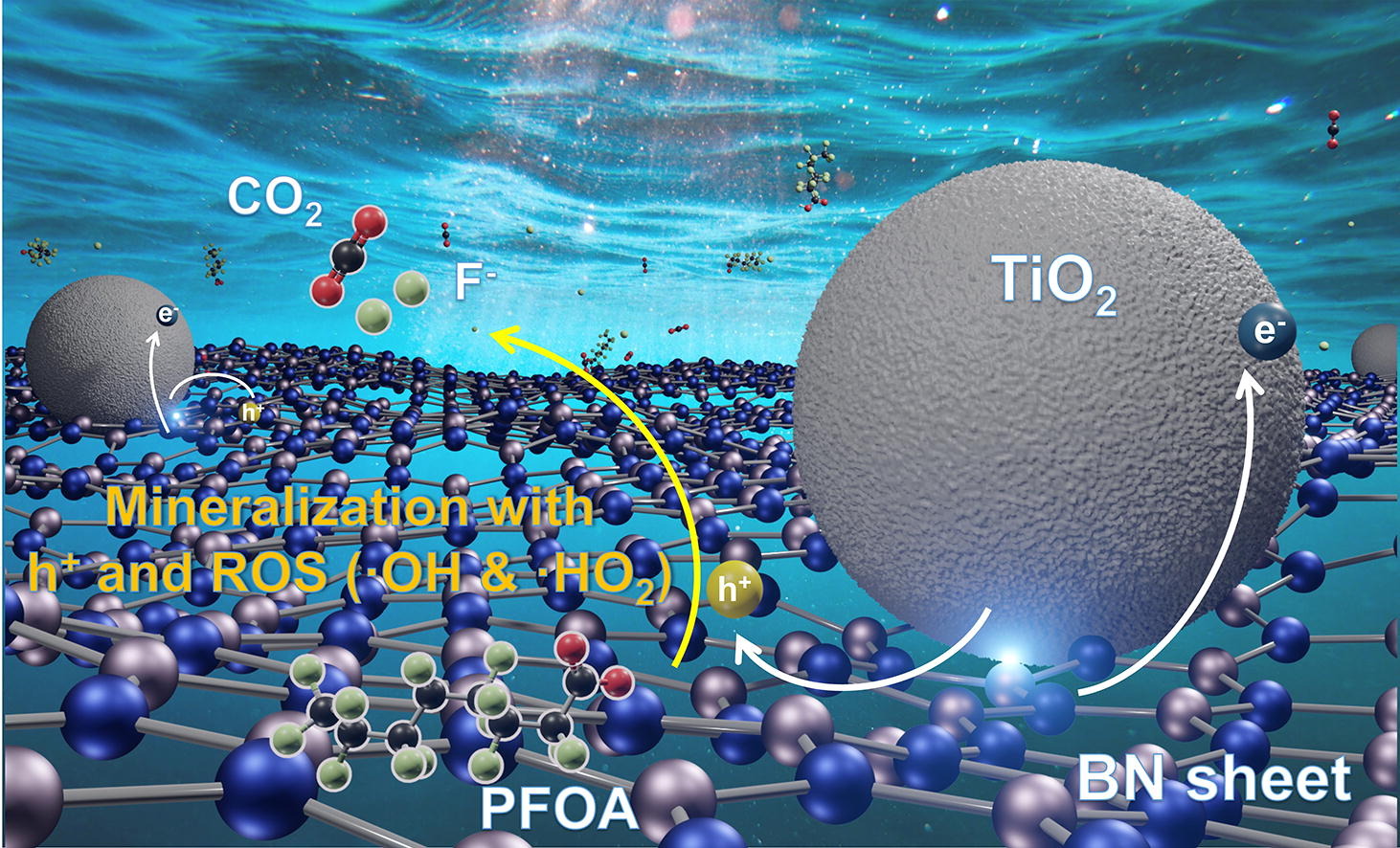
Boron nitride (BN) has the newly-found property of degrading recalcitrant polyfluoroalkyl substances (PFAS) under ultraviolet C (UV-C, 254 nm) irradiation. It is ineffective at longer wavelengths, though. In this study, we report the simple calcination of BN and UV-A active titanium oxide (TiO2) creates a BN/TiO2 composite that is more photocatalytically active than BN or TiO2 under UV-A for perfluorooctanoic acid (PFOA). Under UV-A, BN/TiO2 degraded PFOA ~ 15 × faster than TiO2, while BN was inactive. Band diagram analysis and photocurrent response measurements indicated that BN/TiO2 is a type-II heterojunction semiconductor, facilitating charge carrier separation. Additional experiments confirmed the importance of photogenerated holes for degrading PFOA. Outdoor experimentation under natural sunlight found BN/TiO2 to degrade PFOA in deionized water and salt-containing water with a half-life of 1.7 h and 4.5 h, respectively. These identified photocatalytic properties of BN/TiO2 highlight the potential for the light-driven destruction of other PFAS.
174. Y. Chen, M. Bhati, B.J. Walls, B. Wang, M.S. Wong, and T.P. Senftle "Mechanistic Insight into the Photo-Oxidation of Perfluorocarboxylic Acid over Boron Nitride" Environmental Science & Technology 56, 12, 8942-8952 (2022) DOI: 10.1021/acs.est.2c01637
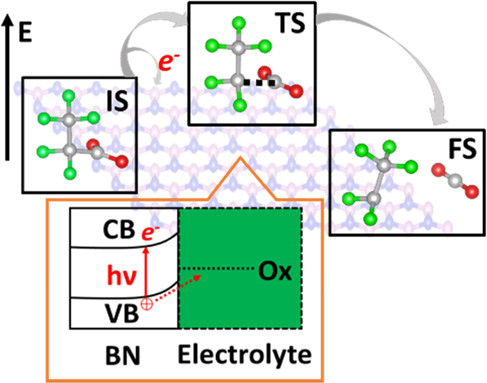
Hexagonal boron nitride (hBN) can photocatalytically oxidize and degrade perfluorocarboxylic acids (PFCA), a common member of the per/polyfluoroalkyl substance (PFAS) family of water contaminants. However, the reaction mechanism governing PFCA activation on hBN is not yet understood. Here, we apply electronic grand canonical density functional theory (GC-DFT) to assess the thermodynamic and kinetic favorability of PFCA photo-oxidative activation on hBN: CnF2n+1COO– + h+ → CnF2n+1· + CO2. The oxidation of all PFCA chains is exothermic under illumination with a moderate barrier. However, the longer-chain PFCAs are degraded more effectively because they adsorb on the surface more strongly as a result of increased van der Waals interactions with the hBN surface. The ability of hBN to act as a photocatalyst is unexpected because of its wide band gap. Therefore, we apply both theoretical and experimental analyses to examine possible defects on hBN that could account for its activity. We find that a nitrogen-boron substitutional defect (NB), which generates a mid-gap state, can enhance UVC (ultraviolet C) absorption and PFCA oxidation. This work provides insight into the PFCA oxidation mechanism and reveals engineering strategies to design better photocatalysts for PFCA degradation.
173. Y. Fang, Z. Zhao, K.N. Heck, L.A. Pretzer, N. Guo, T. Wu, W. Zhang, J.T. Miller, and M.S. Wong "Thermal Annealing Effects on Palladium-decorated Gold Nanoparticle Catalysts" Journal of Catalysis 410, 246-255 (2022) DOI: 10.1016/j.jcat.2022.04.007
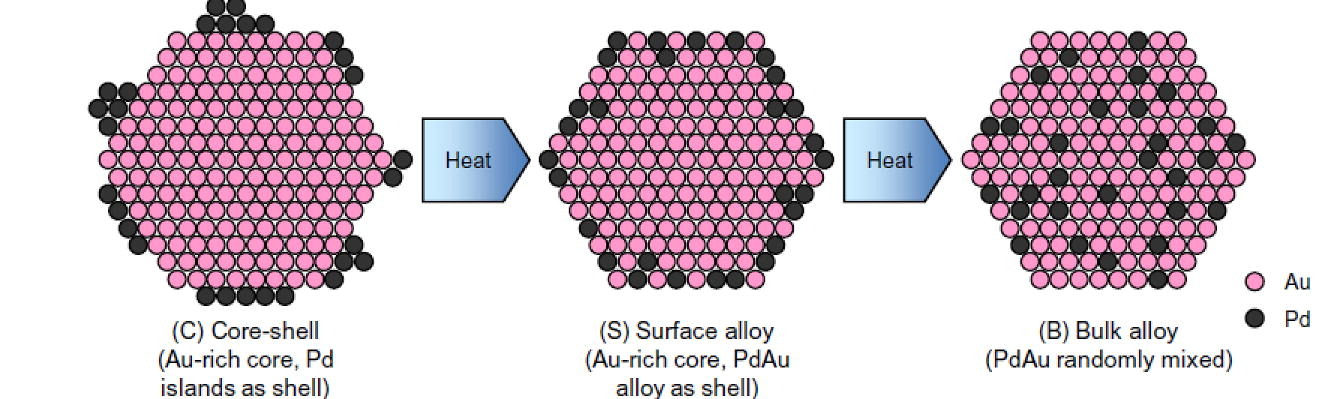
Palladium metal supported on gold in the form of surface ensembles have enhanced catalytic properties compared to monometallic Pd, as exemplified by Pd-decorated Au nanoparticles (Pd-on-Au NPs) for various room-temperature reactions. Whereas the catalytic properties and nanostructure of Pd-on-Au NPs are not known at higher temperatures, this work focuses on thermal annealing effects on the Pd-on-Au NP nanostructure, bimetal distribution, and room-temperature water-phase trichloroethene hydrodechlorination (TCE HDC) as the model reaction. Analysis of the average coordination environment of Pd and Au atoms through x-ray absorption spectroscopy showed that as-synthesized Pd-on-Au NPs transitioned from a Au core/Pd shell structure to Au-rich core/PdAu surface alloy or PdAu mixed alloy structures depending on the Pd surface coverage (30-150 sc%) and annealing temperature (100-400 °C). The HDC activity strongly correlated with Pd ensemble size, where the as-formed Pd islands exhibited one order of magnitude enhanced activity compared to monometallic Pd. Higher annealing temperatures led to a surface/mixed alloy structure with smaller Pd ensemble size, resulting in lower activity but still ~3 times more active than monometallic Pd. These results illustrate the importance of catalyst structure on activity and the usefulness of metal-decorated metal catalysts for higher-temperature reactions.
172. C.L. Conrad, W.C. Elias, S. Garcia-Segura, M.A. Reynolds, and M.S. Wong "A Simple and Rapid Method of Forming Double-Sided TiO2 Nanotube Arrays" ChemElectroChem 9, 7, e202200081 (2022) DOI: 10.1002/celc.202200081
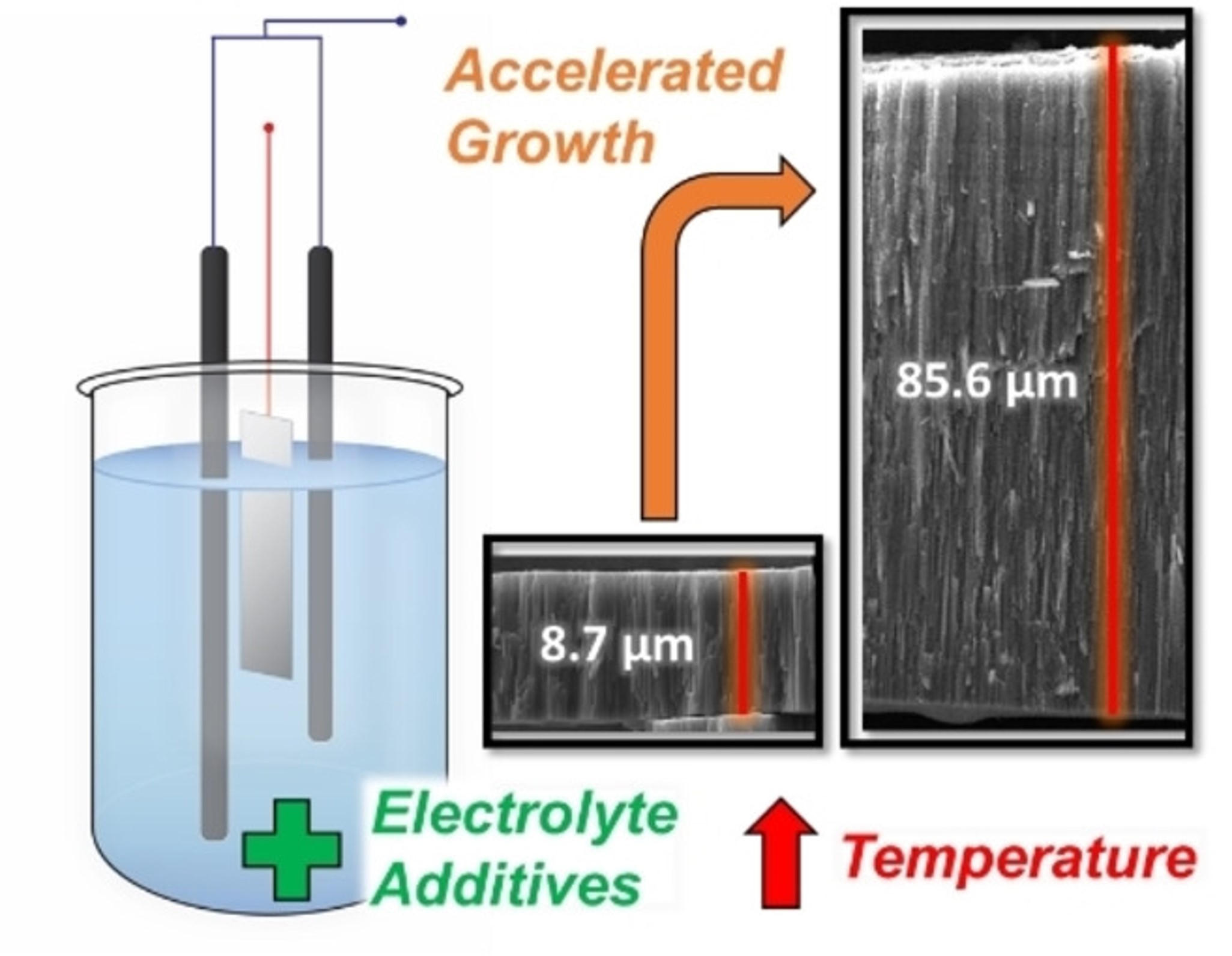
Highly ordered TiO2 nanostructures, known as nanotube arrays (NTAs), exhibit potential in various energy, chemical sensing, and biomedical applications. Owing to its simplicity and high degree of control, titanium anodization serves as the prevailing NTA synthesis method. However, the practicality of this approach is marred by sluggish and inconsistent growth rates, on the order of 10 nm min−1. Growth rates strongly depend on the electrolyte conductivity, yet most reports neglect to consider this property as a measured and controllable parameter. Here, we have systematically determined a broad set of conditions (at 60 V applied potential, elevated temperatures) that allow researchers to fabricate NTAs quickly and simply. By modulating conductivity through variation of bulk electrolyte temperature and the controlled addition of several hydroxy acid species, we achieve consistent accelerated growth up to 10 times faster than traditional methods. We find that regulating the solution conductivity within a desired region (e.g., ~800–1000 μS cm−1) enabled the fabrication of double-sided NTA layers of around 10 μm and 90 μm NTA in 10 and 180 min, respectively.
171. R. Bansal, R. Verduzco, M.S. Wong, P. Westerhoff, and S. Garcia-Segura "Development of nano boron-doped diamond electrodes for environmental applications" Journal of Electroanalytical Chemistry 907, 116028 (2022) DOI: 10.1016/j.jelechem.2022.116028
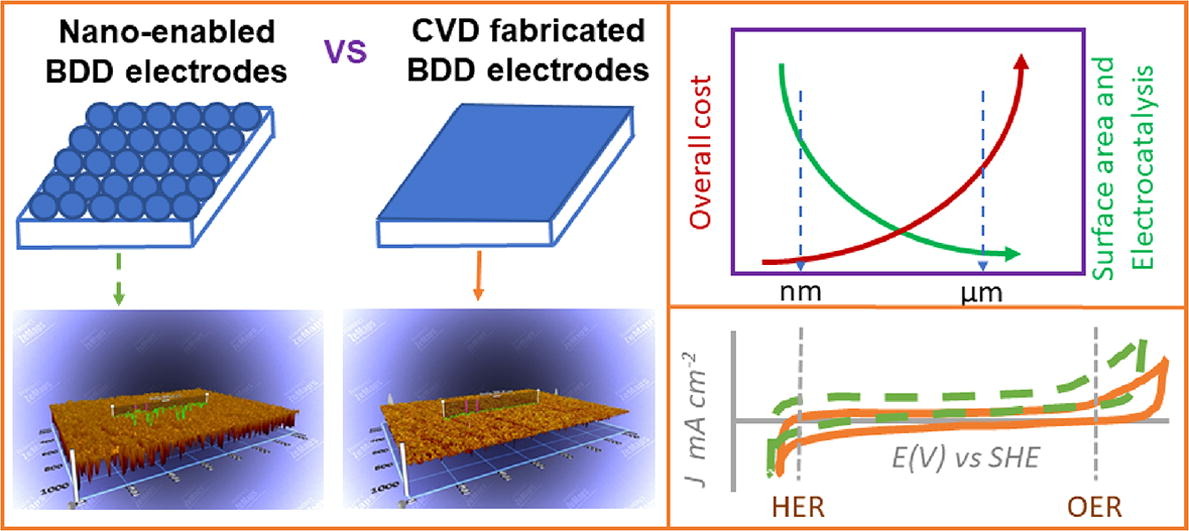
Boron doped diamond (BDD) is an outstanding electrode material with unique electrocatalytic properties and excellent stability, relevant to electrochemical advanced oxidation processes and electroanalytical techniques. From an environmental sustainability viewpoint, BDD electrodes are comprised only of earth abundant elements (carbon, boron, oxygen). However, a major drawback is the high manufacturing costs per unit surface area for BDD electrodes when fabricated using chemical vapor deposition or comparable surface deposition processes. BDD nanoparticles can provide an alternative manufacturing process that reduces costs by over 1000-fold while also improving catalytic activity. Herein, we demonstrate that nano-BDD electrodes can be fabricated by depositing BDD nanoparticles on a silicon substrate using a Nafion® ink-casting method. Scanning electron microscopy (SEM), Raman spectroscopy, and Fourier transform infrared spectroscopy (FTIR) were used to investigate the electrode structural and morphological properties, which were compared to BDD electrodes manufactured using standard methods. Cyclic voltammetry measurements revealed similar electrochemical properties for both electrodes, with a broad “electrochemical window”, essential for effective production of ∙OH radicals without oxygen generation, providing an energy-efficient approach to degradation of pollutants in water. The electrocatalytic properties of the nano-BDD enabled electrodes were investigated using a [Fe(CN)6]3-/4- redox probe. The sensing properties of as-prepared nano-BDD electrodes was studied using Dopamine.
170. S. Guo, H. Li, K.N. Heck, X. Luan, W. Guo, G. Henkelman, and M.S. Wong "Gold boosts nitrate reduction and deactivation resistance to indium-promoted palladium catalysts" Applied Catalysis B: Enviornmental 305, 121048 (2022) DOI: 10.1016/j.apcatb.2021.121048
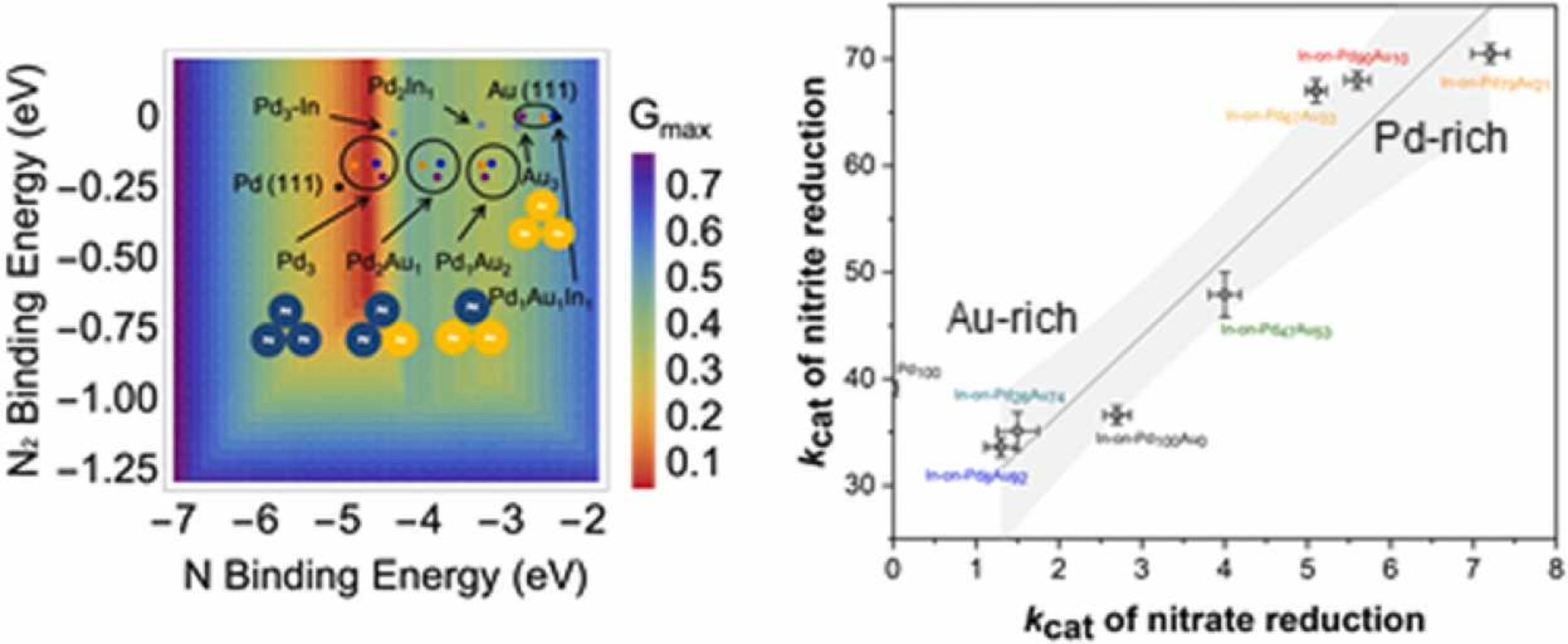
In-decorated PdAu catalysts were synthesized and investigated for the aqueous reduction of nitrate. We first synthesized PdAu nanoparticles (NPs) with varying Pd:Au ratios onto which we deposited In. These In-on-PdAu NPs showed higher activity than In-on-Pd NPs for NO3- (kcat=4.0–7.2 vs 2.7 L/gtotal_(Pd+In)/min) due to electronic and ensemble effects. Au-rich NPs had lower activity (kcat=1.3–1.5 L/gtotal_(Pd+In)/min) due to unfavorable hydrogen and nitrate/nitrite binding energies. In-on-PdAu NPs showed higher N2 selectivity than In-on-Pd NPs (90 +% vs 60% at 40% conversion), and had higher activity for NO2-. Density functional theory calculations suggest that Au weakens metal-N bonding which enables nitrite reduction over Pd. Nitrate reduction is promoted by greater mobility of H-adatoms to regenerate InOx and faster spillover of nitrite onto PdAu. When challenged with species found in drinking water, the trimetallic NPs were more active than bimetallic NPs (1.0–1.6 vs 0.6 L/gtotal_(Pd+In)/min). These results expand the prospects of practical catalytic denitrification.